Every living thing on Earth keeps time. Plants lean toward morning light and close at dusk. Migratory birds depart on cue with the season. The ocean pulses with the tides. Even a single-celled organism responds to the rhythm of day and night. Life, in all its forms, has been shaped by the planet's 24-hour rotation — and the human body is no exception.
Inside each of us, an intricate biological timekeeping system runs continuously, coordinating sleep and wakefulness, hormone release, body temperature, metabolism, and immune defense — all aligned to the cycle of light and dark. Most people experience this as a natural pull toward sleep at night and alertness in the morning, without ever thinking about why.
This internal clock is governed by what scientists call the circadian rhythm — from the Latin circa ("about") and dies ("day"). The term was coined by chronobiologist Franz Halberg in 1957, but the true depth of this system only became clear decades later.
In 2017, the Nobel Prize in Physiology or Medicine was awarded to Jeffrey C. Hall, Michael Rosbash, and Michael W. Young for their discovery of the molecular mechanisms that control circadian rhythms. Their work revealed that nearly every cell in the body carries its own clock — and that disrupting these clocks has profound consequences for human health.

Figure 1.2017 Nobel Prize in Physiology or Medicine awarded for the discovery of circadian rhythm molecular mechanisms
Today, circadian science touches fields as diverse as oncology, metabolic medicine, neurology, and skincare. Yet for most people, the practical implications of their biological clock remain largely invisible — until things go wrong.
This article explains what circadian rhythms are, what throws them off course, who is most at risk, and what the latest science — including emerging research on Urolithin A — reveals about how to protect and restore them.
What Is Circadian Rhythm?
Your body runs on a 24-hour internal clock — and it controls far more than just when you feel sleepy.
Circadian rhythm is a near-24-hour biological cycle that governs sleep, wakefulness, hormone release, metabolism, immune function, and cellular repair. Think of it as an invisible conductor orchestrating every organ and tissue in your body, timing each function to run at its peak moment of the day.
This system has two key layers working together:
The central clock — located in the suprachiasmatic nucleus (SCN) of the hypothalamus. It receives light signals from the retina and sends timing instructions to the rest of the body via hormones like melatonin and cortisol [1].
Peripheral clocks — found in virtually every organ: liver, heart, skin, gut, and muscle. These local clocks take their cues from the SCN and regulate hundreds of rhythm-related genes within their respective tissues [2].
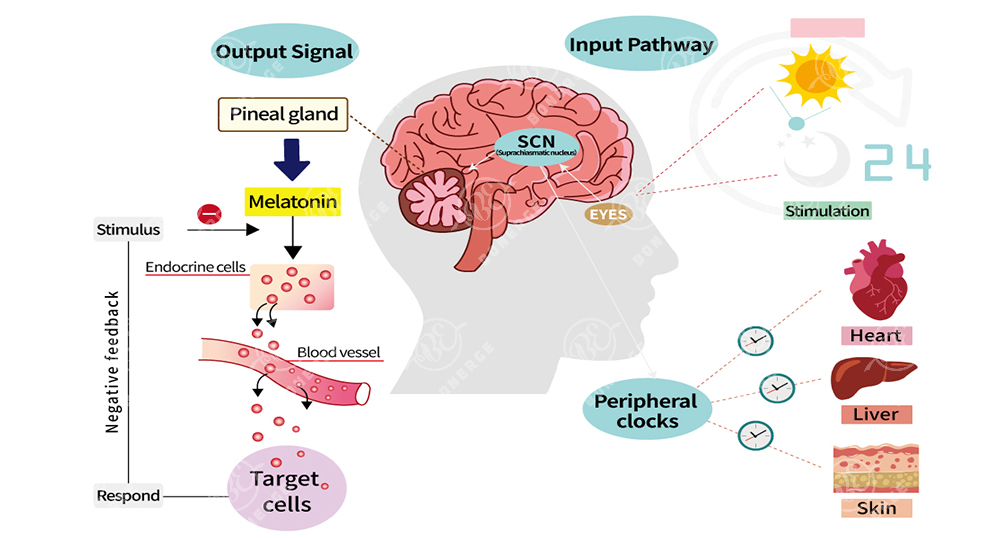
Figure 2.The central and peripheral circadian clock system in humans
How the Sleep/Wake Cycle Works
The sleep/wake cycle is the most visible expression of your circadian rhythm, but it’s only part of the picture.
During waking hours, cortisol peaks in the morning to sharpen focus and energy. As darkness falls, the SCN signals the pineal gland to release melatonin, which lowers core body temperature and prepares the body for sleep. During deep sleep, cells repair, muscles rebuild, proteins and hormones are synthesized, and the brain consolidates memories and clears metabolic waste.
When this cycle runs smoothly, you wake refreshed, think clearly, and feel physically resilient. When it breaks down — even subtly — the consequences spread across every system in the body.
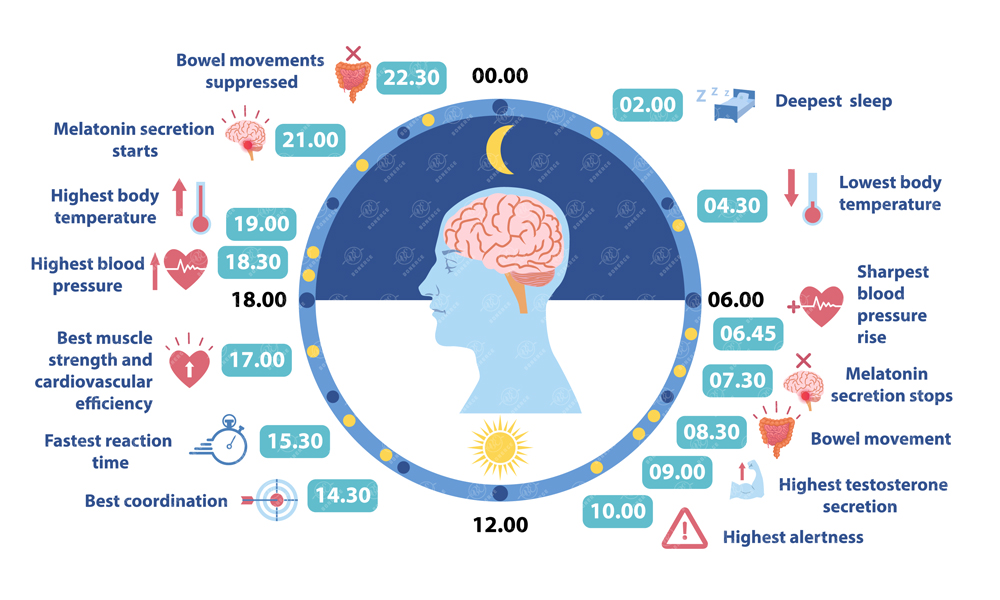
Figure 3.The 24-hour human circadian rhythm timeline
What Disrupts Your Circadian Rhythm?
Modern life is full of circadian disruptors. Understanding them is the first step toward fixing them.
Genetic Factors
Mutations in core clock genes — including BMAL1, CLOCK, PER, and CRY — can directly destabilize biological rhythms. For example, mice lacking BMAL1 show severely disrupted behavioral rhythms, with loss of rhythmic expression in downstream target genes [3]. In humans, mutations in PER2 are linked to Familial Advanced Sleep Phase Syndrome.
Natural Aging
As we age, circadian rhythm amplitude — the strength of the oscillation — gradually weakens. Melatonin and cortisol secretion patterns flatten. Older adults commonly experience a “phase advance,” where sleep and wake times shift earlier by 1–2 hours compared to younger adults. Research also shows that lipid circadian rhythms can lose up to 14% of their amplitude with age, directly elevating risks for metabolic dysfunction and neurodegenerative disease [4].
Light Exposure at the Wrong Time
Light is the most powerful zeitgeber (time-giver) for the SCN. Nighttime light exposure — especially from screens — is particularly damaging. Exposure to 500–600 lux or more at night disrupts digestive and metabolic rhythms, impairs insulin sensitivity, and causes abnormal post-meal glucose and insulin responses [5, 6].
Irregular Sleep Timing
Even if you sleep eight hours, when you sleep matters. Studies show that a 12-hour shift in sleep phase (sleeping days, waking nights) worsens average daily blood glucose by 6% and insulin levels by 22% [7].
Irregular Meal Times
The gut has its own peripheral clock. Delaying dinner by five hours for six consecutive days shifts the blood glucose rhythm by approximately 5.7 hours [8]. Moving lunch from 1:00 PM to 4:30 PM raises the glucose AUC in tolerance tests by 46% and reduces fasting carbohydrate oxidation [9].
Who Is Most at Risk for Circadian Rhythm Disruption?
Certain groups face a higher baseline risk of sleep/wake cycle dysregulation:
Shift workers experience a chronic conflict between their circadian clock and their work schedule. This population shows near-universal clock gene rhythm delays of 2.5–3 hours. Critically, shift work involving circadian disruption is classified as a Group 2A carcinogen by the International Agency for Research on Cancer.
Frequent travelers crossing time zones face acute circadian misalignment, with eastward travel typically causing greater disruption than westward.
Night-owl or late-night screen users who habitually delay sleep push their circadian phase later, accumulating a “social jet lag” between their biological clock and their schedule.
Older adults deal with a gradually weakening clock, increased phase advance, and reduced melatonin output — all of which compound sleep fragmentation.
People who are blind often lack the light input needed to entrain the SCN, making them susceptible to non-24-hour sleep/wake disorder.
The stakes are real. A large prospective cohort study found that people with regular sleep patterns had [10]:
16–39% lower cancer-related mortality risk
22–57% lower cardiovascular and metabolic disease mortality
20–48% lower all-cause mortality compared to those with the most irregular sleep
The Health Consequences of Circadian Rhythm Disruption
Circadian misalignment doesn’t just affect sleep. It triggers a cascade of downstream consequences.
Metabolic Dysfunction
Circadian disruption directly impairs insulin secretion timing and glucose/lipid metabolism. Clock gene mutations (BMAL1, CLOCK) in animal models reduce hepatic glycogen synthesis, lower fatty acid oxidation rates, and rapidly produce insulin resistance and fatty liver [11]. In humans, a Mediterranean population study found that naps exceeding 30 minutes were associated with higher BMI, waist circumference, blood pressure, and fasting blood glucose — with a metabolic syndrome score 8.1% higher than non-nappers [12].
Cognitive Decline and Neurodegeneration
Long-term prospective studies (5–41 years of follow-up) consistently show that people with disrupted rhythms — particularly shift workers and older adults — experience faster cognitive decline and significantly higher rates of Alzheimer’s and Parkinson’s disease [13]. The proposed mechanism involves impaired cerebral blood flow, reduced neuronal repair, and accelerated accumulation of amyloid-β in the brain.
Skin Aging and Impaired Wound Healing
Skin cell repair, immune response, and collagen synthesis are all circadian-gated. Clinical data show that UVB irradiation at night causes more pronounced erythema than the same dose in the morning, correlating with changes in the rhythm protein CRY2 (p=0.015) [14]. Analysis of a large burn injury database found that nighttime burns heal approximately 60% slower than daytime burns [15].
Muscle Loss
In animal models, deletion of CLOCK or BMAL1 genes reduces skeletal muscle tone by ~30% and shrinks mitochondrial volume by ~40% [16]. In humans, chronic circadian misalignment — from shift work or persistent sleep disorders — significantly increases the risk of sarcopenia and osteoporosis.
Immune Suppression and Chronic Inflammation
Gut microbiota composition follows a circadian pattern. Disruption reduces populations of beneficial bacteria like Faecalibacterium prausnitzii and Agathobacter — producers of short-chain fatty acids critical for gut barrier integrity. The result: weakened immune defenses, elevated chronic inflammation, and increased susceptibility to metabolic and autoimmune conditions.

How to Reset Your Circadian Rhythm: Evidence-Based Strategies
Resetting a disrupted circadian rhythm requires consistency across multiple behavioral domains. Here’s what the science supports:
1. Anchor Your Light Exposure
Get 10–15 minutes of natural morning light within one hour of waking — even on cloudy days. This suppresses residual melatonin and sends a clear “daytime” signal to the SCN.
At night, avoid bright screens 1–2 hours before bed. Use blue-light filtering glasses or software. Keep the bedroom as dark as possible.
2. Set a Fixed Sleep/Wake Schedule
This is the single most powerful habit. Wake up and go to sleep at the same time every day — including weekends. Aim for 7–9 hours. The body’s clock depends on predictability. Inconsistency undoes even good sleep hygiene.
3. Time Your Meals Strategically
Eat at consistent times each day, with breakfast and lunch as your larger meals. Finish dinner at least 2–3 hours before bed. Avoid late-night eating, especially high-fat or high-sugar foods. Avoid caffeine and alcohol in the evening — both fragment sleep architecture.
4. Move at the Right Time
Morning or afternoon outdoor exercise reinforces circadian signals and improves sleep quality. Avoid vigorous exercise within 3 hours of bedtime, as it raises core body temperature and delays sleep onset.
5. Optimize Your Sleep Environment
Keep your bedroom cool (ideally 18–20°C / 64–68°F). Darkness and quiet matter. Use the bed only for sleep — strengthening the mental association between bed and rest.
6. Manage Stress and Pre-Sleep Wind-Down
Cortisol dysregulation from chronic stress directly disrupts melatonin timing and sleep structure. Build a consistent wind-down routine: light reading, meditation, deep breathing, or a warm bath. Avoid stimulating content or stressful conversations in the final hour before bed.
7. Consider Targeted Nutritional Support
Specific nutrients have demonstrated roles in circadian regulation. These include vitamin B2, B6, folate, vitamin D3, tryptophan, and — increasingly supported by emerging research — Urolithin A. Supplementation should be guided by individual needs and, where appropriate, professional advice.
Urolithin A and Circadian Rhythm: Emerging Science
Among nutritional compounds now being studied for circadian support, Urolithin A (UA) has attracted significant scientific attention.
How Urolithin A Supports the Biological Clock
Gut-to-Brain Clock Synchronization
Circadian rhythms play a critical role in maintaining gut homeostasis and immune function. Research shows that Urolithin A regulates core clock gene expression in gut epithelial cells, restoring desynchronization between clock genes and tight junction proteins caused by inflammation. In animal models, UA restored normal expression rhythms of both tight junction proteins (Cldn1, Cldn4) and clock genes (BMAL1, PER2) in colonic tissue, while simultaneously modulating the central clock in the SCN [17].
This gut-to-brain axis is a key mechanism: by restoring peripheral clock function in the gut, Urolithin A helps re-anchor the entire circadian hierarchy.
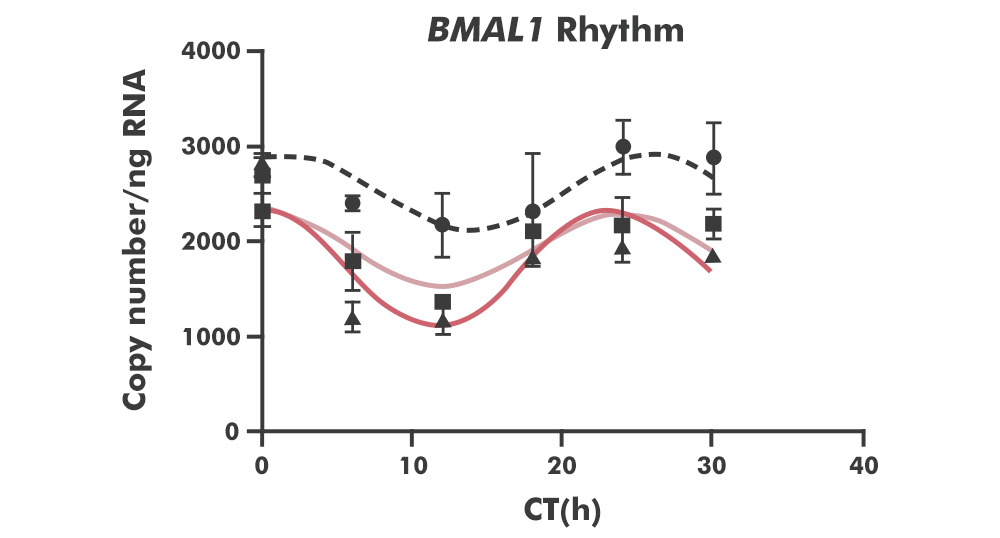
Figure 4.BMAL1 circadian expression rhythm in different treatment groups
Restoring Rhythm Amplitude in Aging Cells
Aging impairs the temporal precision of the circadian clock — oscillations slow down and weaken. In senescent cell models (TIG-3), Urolithin A dose-dependently enhanced BMAL1 promoter-driven luciferase rhythm amplitude by up to 4fold, demonstrating meaningful functional restoration of circadian regulation in aged cells [18].
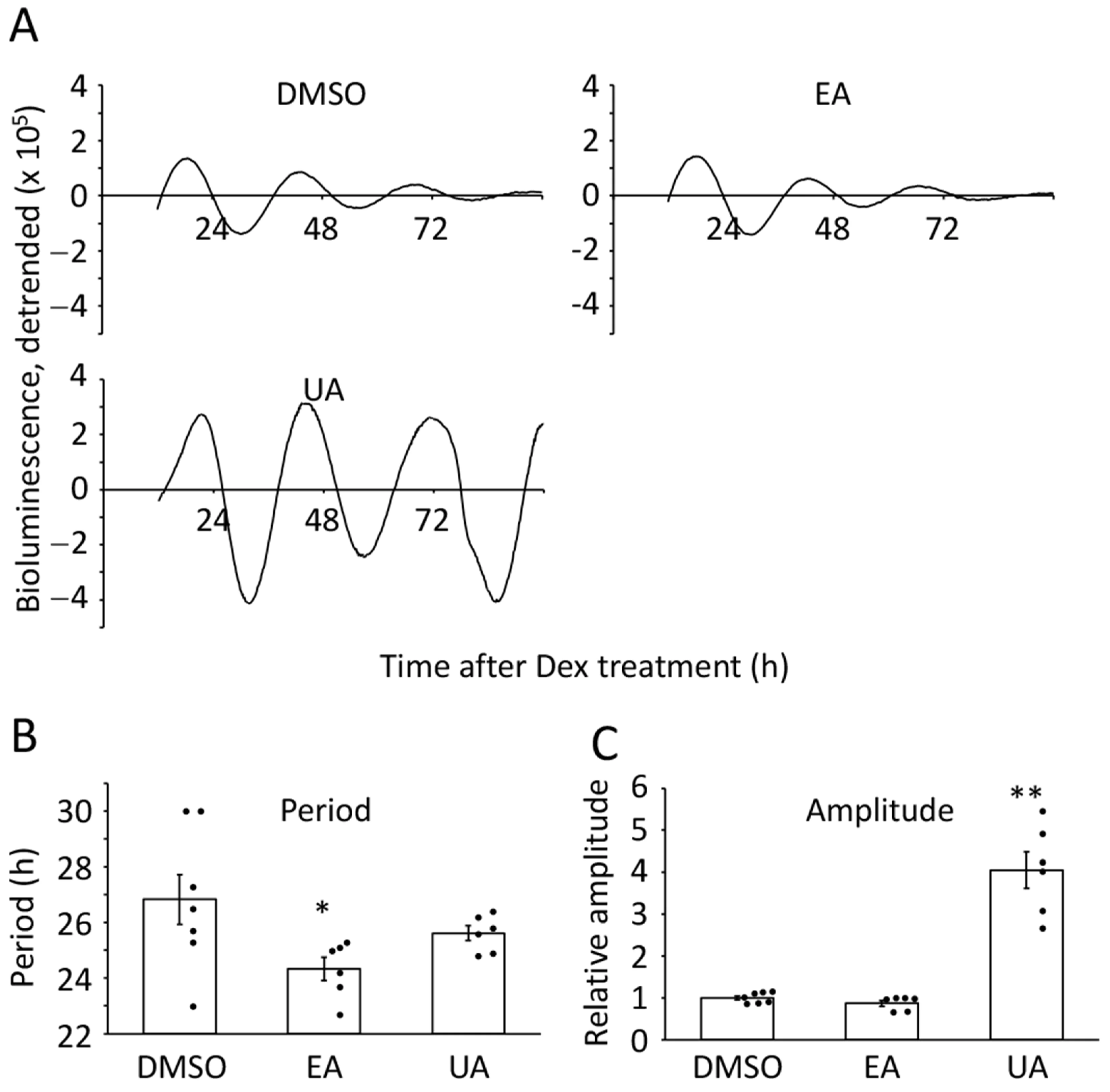
Figure 5.Urolithin A enhances circadian rhythm amplitude in senescent cells
Boosting NAD⁺ for Daytime Energy
NAD⁺ is a key metabolic cofactor that declines with age and with circadian disruption. Urolithin A significantly elevates cellular NAD⁺ levels through activation of the SIRT1-NAMPT pathway. In mouse studies, UA increased NAD⁺ levels by 50%, showing approximately 5× the efficacy of nicotinamide riboside (NR) at equivalent doses [19]. Higher NAD⁺ supports mitochondrial energy metabolism and is directly linked to improved daytime vitality and cognitive performance.
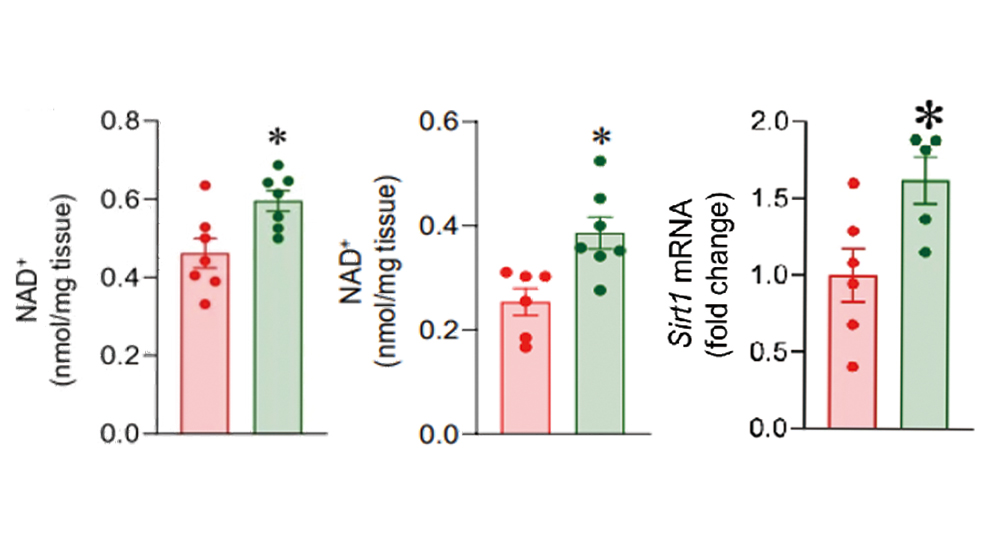
Figure 6.Urolithin A elevates cellular NAD⁺ levels and upregulates Sirt1 expression
Fighting Sleep-Deprivation Fatigue
In sleep-deprived mouse models, Urolithin A outperformed caffeine on key performance markers:
35% improvement in grip strength (vs. 12% with caffeine)
100% prolongation of fatigue resistance on the rotarod test
Concurrent improvement in gut microbiota imbalance [20]
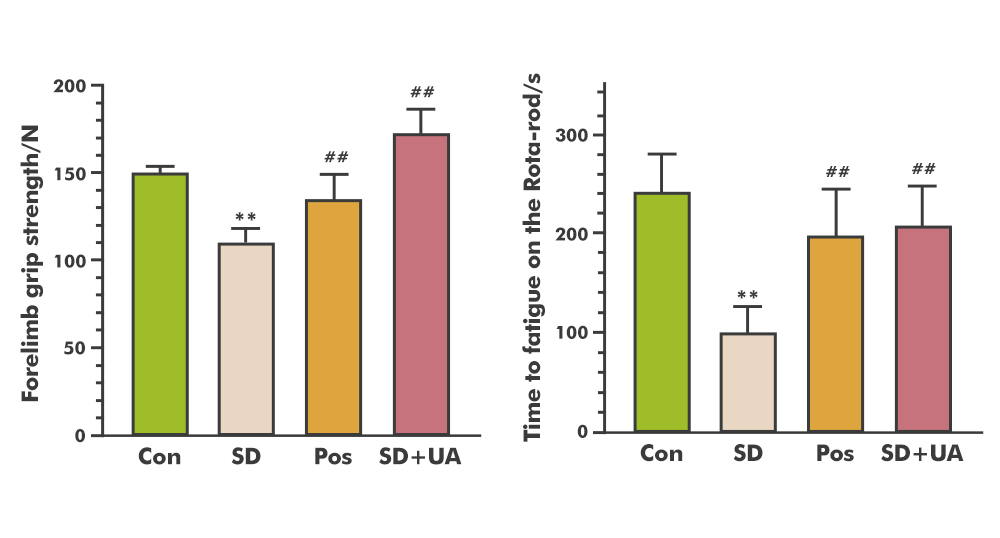
Figure 7.Urolithin A (UA) improves physical performance in sleep-deprived (SD) mouse models
Supporting Skin Repair During Sleep
During the nighttime repair cycle, Urolithin A enhances BMAL1 rhythm amplitude in dermal fibroblasts, upregulating collagen synthesis genes (including COL1A1) to promote skin barrier repair and elastic fiber regeneration. This creates a measurable bridge between circadian health and skin quality — the mechanism behind the concept of “beauty sleep.”
Application Potential
Urolithin A’s multi-axis approach — gut clock restoration, cellular rhythm amplification, NAD⁺ enhancement, and anti-fatigue effects — positions it as a compelling candidate for circadian rhythm supplements targeting healthy aging, metabolic wellness, and sleep/wake cycle support.
Bonerge’s StanYouth® Urolithin A provides ultra-pure, standardized Urolithin A for nutraceutical and supplement formulations, backed by manufacturing precision and third-party quality verification.
References
[1] Panda S, et al. Cell, 2002.
[2] Moore GWK, et al. Nature Communications, 2021.
[3] Kondratov RV, et al. Genes & Development, 2006.
[4] Kuatov R, et al. Nutrients, 2025; 17(1):20.
[5] Albreiki MS, et al. Endocrine Connections, 2017.
[6] Gil-Lozano M, et al. Am J Physiol Endocrinol Metab, 2016.
[7] Scheer FA, et al. PNAS, 2009.
[8] Wehrens SMT, et al. Current Biology, 2017.
[9] Bandin C, et al. Int J Obes (Lond), 2015.
[10] Windred DP, et al. Sleep regularity is a stronger predictor of mortality risk than sleep duration: A prospective cohort study. Sleep, 2024; 47(1):zsad253.
[11] Tong X, Yin L. Compr Physiol, 2013; 3(2):917-40.
[12] Vizmanos B, et al. Obesity (Silver Spring), 2023.
[13] Leng Y, et al. Lancet Neurol, 2019.
[14] Nikkola V, et al. J Invest Dermatol, 2018.
[15] Hoyle NP, et al. Sci Transl Med, 2017.
[16] Andrews JL, et al. PNAS, 2010.
[17] Du Y, et al. Effect of Urolithin A on the Improvement of Circadian Rhythm Dysregulation in Intestinal Barrier Induced by Inflammation. Nutrients, 2024; 16(14):2263.
[18] Kuatov R, et al. Urolithin A Modulates PER2 Degradation via SIRT1 and Enhances the Amplitude of Circadian Clocks in Human Senescent Cells. Nutrients, 2024; 17(1):20.
[19] Ghosh N, et al. Urolithin A augments angiogenic pathways in skeletal muscle by bolstering NAD+ and SIRT1. Sci Rep, 2020; 10(1):20184.
[20] Zhu H, et al. Urolithin A Ameliorates Athletic Ability and Intestinal Microbiota in Sleep Deprivation from the Perspective of the Gut-Muscle Axis. Mol Nutr Food Res, 2024; 68(7):e2300599.




































 Time:Apr 02, 2026
Time:Apr 02, 2026
 Share:
Share:










 +1(425)866-9043
+1(425)866-9043
 sales@bonerge.com
sales@bonerge.com