Menopause is more than a transition—it is a cardiovascular tipping point. While many women focus on immediate symptoms like hot flashes or bone density, a far more silent threat is often overlooked.
"Although many people consider breast cancer to be the No. 1 killer of women in the U.S., heart disease kills more women than all forms of cancer combined,"
says Dr. Garima Sharma, a volunteer for the American Heart Association (AHA). The statistics are sobering: while 1 in 39 women dies from breast cancer each year, 1 in 3 dies from cardiovascular disease (CVD). That equates to approximately one death every minute—and menopause plays a central role in this escalating risk.[1]
Dr. Sharma emphasizes that the years leading up to and during menopause are a "critical window" for understanding and addressing these risks. For women in their 40s and 50s, the hormonal and physiological shifts are not just life stages—they are signals that the body's internal protective mechanisms are changing.[1]
For decades, this rise in risk was attributed primarily to the decline of estrogen, which helps maintain vascular flexibility and reduce inflammation. As estrogen levels drop, the heart’s natural "shield" weakens. However, recent clinical evidence suggests the story is more complex—and involves a less visible but equally vital factor: metabolic changes linked to the gut microbiome.

Beyond Estrogen: Gut Metabolism and TMAO in Menopause
Recent clinical findings indicate that postmenopausal women exhibit higher levels of trimethylamine N-oxide (TMAO)—a metabolite associated with arterial stiffness and atherosclerosis—compared with premenopausal women and age-matched men.[2]
Counterintuitively, these increases occur even when dietary intake of TMAO precursors (such as choline and carnitine) is low. This suggests that menopause alters TMAO metabolism itself, rather than just reacting to diet. The decline in estrogen can disrupt the gut microbiota, creating conditions for TMAO to increase—a "hidden threat" to cardiovascular health that exists alongside more visible symptoms like hot flashes or bone loss.
Why Diet Alone May Not Protect Heart Health After Menopause
One of the most important concepts in modern nutrition is individual variability. Not everyone can convert healthy foods into heart-protective compounds. This ability depends entirely on your gut microbiome—a biological signature researchers call "metabotypes."
For instance, while some people can naturally produce Urolithin A or S-equol from plants, many others lack the specific gut bacteria to do so. This is why two people can eat the same healthy diet but see completely different health results.[3]
New Clinical Evidence: Polyphenols Lower TMAO in Postmenopause
Emerging clinical research reveals how your unique gut microbiome translates dietary nutrients into heart-protective outcomes—especially for postmenopausal women.
In a high-quality, randomized, placebo-controlled crossover trial (n = 90), healthy postmenopausal women supplemented with a polyphenol complex derived from pomegranate, Polygonum cuspidatum (Japanese Knotweed), and red clover. These plant extracts are natural precursors to Urolithin A and Equol, compounds known for their cardioprotective properties.[4]
The results were striking: the intervention significantly reduced levels of urinary TMAO and dimethylamine (DMA)—two key biomarkers linked to cardiovascular risk. However, the study found that these benefits are not "one-size-fits-all." The reduction in heart-clogging TMAO was most significant in specific "metabotypes":
UM-A (Urolithin A-producers): Those whose gut bacteria efficiently process pomegranate ellagitannins.
EP (Equol-producers): Women capable of converting soy/clover isoflavones into equol.
LNP (Lunularin-non-producers): A specific microbial profile related to resveratrol metabolism.
This research reinforces that managing heart health after menopause requires a precision nutrition approach, as the effectiveness of polyphenols depends heavily on your individual gut ecosystem.
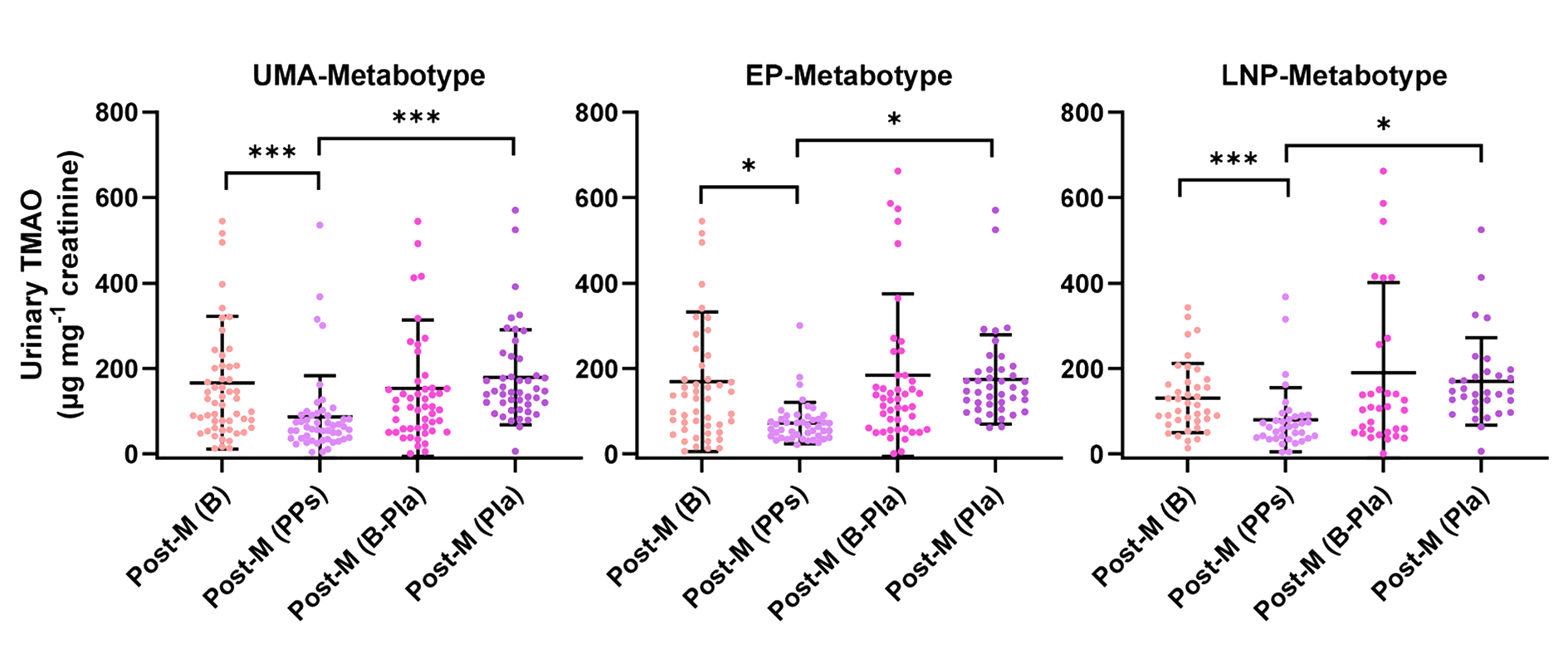
Figure 1. Changes in urinary TMAO levels in postmenopausal women (Post-M). Post-M (B) represents baseline levels; Post-M (PPs) represents levels after polyphenol intervention; Post-M (B-Pla) represents the baseline levels for the placebo group; and Post-M (Pla) represents levels after placebo administration
How Urolithin A and S-equol Support Cardiovascular Function
The findings from the recent clinical trial in Spain align with a growing body of evidence. Multiple human and preclinical studies now confirm that Urolithin A and S-equol provide significant, measurable benefits for heart and vascular function.
Rather than acting alone, these two gut-derived metabolites work through complementary pathways to address the root causes of cardiovascular decline.
At the mechanistic level, their benefits are largely driven by antioxidant and anti-inflammatory activities, particularly in protecting vascular endothelial function and modulating TMAO-related effects.
Both Urolithin A and S-equol possess potent antioxidant and anti-inflammatory properties. Their primary value lies in their ability to target vascular endothelial damage. Specifically, they help counteract the negative effects of TMAO, a metabolic toxin that can impair blood vessel function. By neutralizing this tension, they support the integrity of the arterial walls.
Urolithin A: Reducing Key Risk Markers
Urolithin A (UA) has been extensively studied for its role in metabolic health. High-quality clinical data highlights its effectiveness:
Managing Ceramides: A four-month study involving healthy older adults demonstrated that UA supplementation significantly lowers levels of ceramides. These lipids are critical indicators of atherosclerosis, metabolic syndrome, and other adverse cardiac events.[5]
Improving Muscle Efficiency: Research in aging and heart failure models shows that UA improves the heart’s ability to contract and relax. This suggests that UA can directly enhance myocardial performance, helping the heart function more efficiently under stress.[6]
S-equol: Strengthening the Body’s Antioxidant Defense
S-equol offers a different layer of protection by interacting with the body's internal systems:
Gene Activation: S-equol promotes the expression of endogenous antioxidant genes. This increases the body’s innate capacity to manage oxidative stress, which is a leading cause of damage to blood vessels and heart tissue.
Preventative Support: By reducing the cumulative impact of oxidative stress, S-equol provides a sustainable way to maintain cardiovascular resilience, particularly for postmenopausal women who may face increased heart health risks.[7]
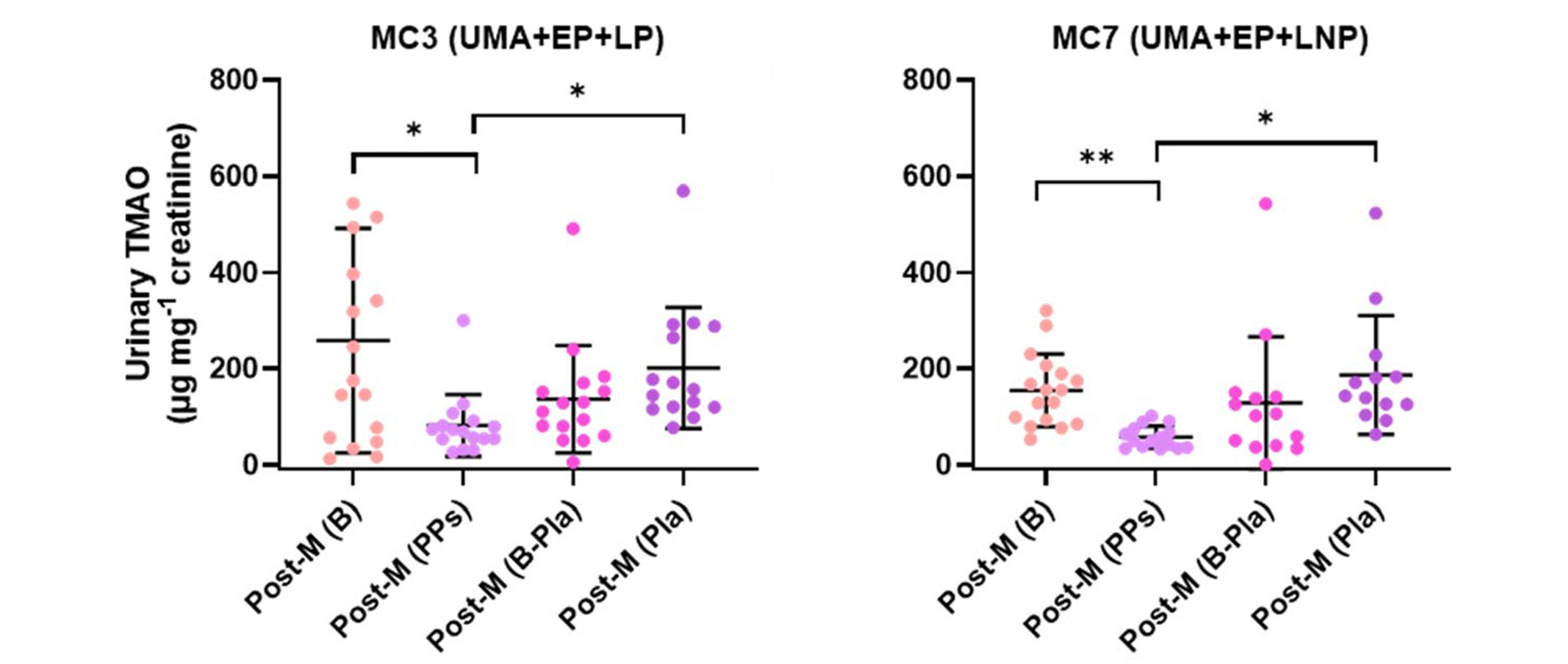
Figure 2. Changes in urinary TMAO levels following polyphenol (PPs) supplementation in postmenopausal women (Post-M), stratified by metabolic clusters (MC3 and MC7).
Precision Nutrition for Menopause: Targeting Gut Metabolism
These insights align with a broader shift in the nutraceutical industry: the move toward precision nutrition.
Direct supplementation of bioactive compounds, such as urolithin A and S-equol, can bypass individual metabolic limitations, ensuring more consistent physiological benefits.
For B2B brands and formulators, these findings open up new opportunities:
Targeted menopause solutions that go beyond symptom relief
Metabolism-focused formulations addressing underlying pathways
Combination strategies leveraging multiple bioactives
Differentiation through science-backed personalization concepts
Rather than focusing solely on hormonal balance, the next wave of innovation may lie in metabolic modulation and gut-derived bioactives.
Future Strategies for Postmenopausal Heart Health
Menopause-related health challenges cannot be addressed through a single pathway. Hormones, metabolism, and the gut microbiome interact in complex ways, and the benefits of polyphenols like urolithin A and S-equol depend on an individual’s metabolic capacity.
For women who cannot naturally produce these metabolites, diet alone is often insufficient. Direct supplementation with EquoYouth® S-equol and StanYouth® urolithin A provides a reliable, efficient, and science-backed way to support cardiovascular and overall health.


This “golden combination” works through metabolism-dependent mechanisms to precisely lower TMAO levels, protect vascular function, and promote sustainable health outcomes.
By delivering the right metabolites to the right people, it ensures comprehensive, personalized, and long-term benefits for women navigating the postmenopausal transition.
References
[1] American Heart Association. Hot news flash: Menopause can impact a woman’s heart health. News release. 2024 Oct 16. https://newsroom.heart.org/news/hot-news-flash-menopause-can-impact-a-womans-heart-health
[2] Battillo DJ, Malin SK. Trimethylamine N-oxide is elevated in postmenopausal women relative to age-matched men and premenopausal women among individuals with obesity. Exp Physiol. 2025; Advance online publication. https://doi.org/10.1113/EP092550
[3] Liu H, Jia K, Ren Z, et al. PRMT5 critically mediates TMAO-induced inflammatory response in vascular smooth muscle cells. Cell Death Dis. 2022;13:299. https://doi.org/10.1038/s41419-022-04719-7
[4] Jarrín-Orozco MP, García-Nicolás M, Romo-Vaquero M, et al. Precision health targeting TMAO in postmenopausal women: Polyphenol effects modulated by urolithin A and equol metabotypes in a randomized, placebo-controlled crossover trial. Food Funct. 2026;17:2037–2049. https://doi.org/10.1039/d5fo05461c
[5] Selma MV, González-Sarrías A, Salas-Salvadó J, et al. The gut microbiota metabolism of pomegranate or walnut ellagitannins yields two urolithin-metabotypes correlating with cardiometabolic risk biomarkers. Clin Nutr. 2018;37:897–905. https://doi.org/10.1016/j.clnu.2017.03.012
[6] Liu S, Faitg J, Tissot C, et al. Urolithin A provides cardioprotection and mitochondrial quality enhancement preclinically and improves human cardiovascular health biomarkers. iScience. 2025;28:111814. https://doi.org/10.1016/j.isci.2025.111814
[7] Zhang T, Liang X, Shi L, et al. Estrogen receptor and PI3K/Akt signaling pathway involvement in S-(-)equol-induced activation of Nrf2/ARE in endothelial cells. PLoS One. 2013;8:e79075. https://doi.org/10.1371/journal.pone.0079075




































 Time:Feb 18, 2026
Time:Feb 18, 2026
 Share:
Share:










 +1(425)866-9043
+1(425)866-9043
 sales@bonerge.com
sales@bonerge.com