If you’re interested in soy isoflavones or natural supplements, you may have heard of S-equol. But what exactly is it? Is it safe to use? This article covers everything you need to know about S-equol—from its basic details to its safety profile. It includes all scientific references and uses the latest scientific research and clinical data to give you a full, accurate overview.

What Is S-Equol? Basic Information
S-equol (S-Equol) is a natural substance, a major byproduct of soy isoflavones—especially daidzein—broken down by microbes in the body. Its chemical name, from the IUPAC, is (3S)-7-hydroxy-3-(4-hydroxyphenyl)-2,3-dihydrochromen-4-one [1]. Unlike daidzein (the substance it comes from), S-equol is more biologically active and easier for the body to use, making it the key active form of soy isoflavones in humans.
It has a CAS number of 531-95-3, a molecular formula of C₁₅H₁₄O₃, and a molecular weight of 242.27 g/mol (see Figure 1 for its chemical structure). This molecular structure gives S-equol unique physical and chemical traits, as well as specific biological effects related to estrogen receptors.
Structurally, S-equol has a chromen-7-ol (dihydropyran) core linked to a phenolic ring, with a unique “chiral center” at the C3 position. This means it has two forms (enantiomers): S-equol and R-equol [1]. The chiral center is very important for its biological activity, as it determines how well the compound can attach to specific receptors in the body.
In humans, only S-equol is made by bacteria in the intestines—specifically, certain types of gut bacteria that can break down daidzein into this active form. The other form, R-equol, is rarely made in the human body and has little to no biological activity [1], which is why not everyone gets the same benefits from soy isoflavones—your gut bacteria play a key role.
Physically, S-equol is a white to off-white (or cream-colored) powder that is non-polar, meaning it does not dissolve well in water [2]. This affects how it is absorbed, but it is still much easier for the body to use than daidzein. It is unstable in acidic conditions and breaks down easily, so it needs to be stored in a stable environment, and its melting point is about 189–190°C [2]—a trait that helps scientists purify and identify it in labs.
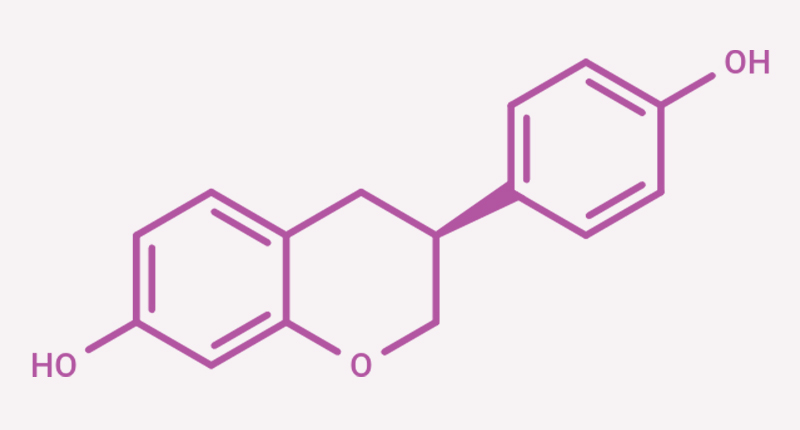
Figure 1. 2D Structure of S-equol
How Is S-Equol Formed and Metabolized in the Body?
Biosynthesis in the Body
Equol was first found in the urine of pregnant mares in 1932 by Marrian and Haslewood [1]. Fifty years later, in the 1980s, scientists found it in human urine too, realizing it is a key byproduct of soy isoflavones—marking the start of research into how it affects human health [1]. Since then, many studies have focused on how it is made and how the body processes it.
As mentioned, equol has two forms: R-equol and S-equol, and only S-equol is naturally made by intestinal bacteria in humans [2]. This process depends on specific gut bacteria that have the right enzymes to turn daidzein into S-equol, a step that decides whether a person can get the biological benefits of soy isoflavones.
Not everyone can make S-equol—this ability is called the “Equol producer phenotype.” Studies show that 20–35% of adults in Western countries can make it after eating soy foods or pure isoflavone supplements [3], while in Asian populations, this number goes up to 50–60% [3]. Experts think this difference comes from diet (more soy intake) and differences in gut bacteria between groups of people.
People who cannot make S-equol (called non-producers) usually break down soy isoflavones into O-desmethylangolensin (O-DMA), a byproduct with no estrogenic activity that does not have the same biological effects as S-equol [4,5]. This difference is important for understanding how different people react to soy-based supplements and diets.
Most natural soy isoflavones (about 97–98%) are in an inactive, glycosylated form in soybeans and soy products—meaning they are bound to sugar molecules [6,7]. These inactive forms cannot be directly absorbed by the human body; they need to be broken down into “aglycones” (free forms) by digestive enzymes before the body can absorb them [6,7].
Take daidzin (a major inactive soy isoflavone) as an example: it is first broken down in the small intestine by β-glucosidase enzymes—made by both the body and gut bacteria—into daidzein, the active free form. Then, cells in the intestine absorb daidzein into the bloodstream [6,7].
After absorption, the liver processes daidzein through “phase II metabolism,” turning it into water-soluble glucuronide or sulfate conjugates that are then excreted in urine or feces, or released into bile and sent back to the intestines [6,7]. These conjugates can be broken down again (deconjugated) in the lower intestine by bacterial enzymes, allowing free daidzein to be reabsorbed and enter “enterohepatic circulation”—a process that makes daidzein (and its byproducts) stay in the body longer [6,7].
Some daidzin or its conjugates reach the colon without being absorbed in the small intestine, where bacterial enzymes break down the glycosylated, sulfated, and glucuronidated forms into free daidzein [8]. Bacteria in the colon then turn this free daidzein into dihydrodaidzein through “reductive reactions,” and the type of bacteria present (such as Lactococcus garvieae or Slackia isoflavoniconvertens) determines whether dihydrodaidzein is turned into active S-equol or inactive O-DMA [8]—the final step that decides if a person is an Equol producer or non-producer.
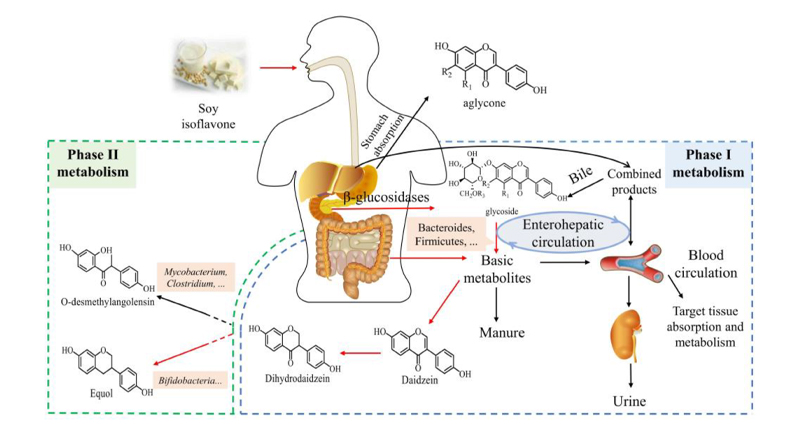
Figure 2. In Vivo Metabolic Pathway of Soy Isoflavones (Daidzin) [2]
In Vivo Catabolism
In the body, S-equol is mainly processed through phase II metabolism, which involves “conjugation reactions” (glucuronidation and sulfation) rather than phase I oxidation. Most of its byproducts are excreted in urine, while some are released into bile to enter enterohepatic circulation—helping keep S-equol levels stable in the blood [9].
S-equol has hydroxyl groups only at the 4′ and 7 positions of its molecular structure, which are the only places where glucuronidation or sulfation can happen [10]. This means the byproducts of S-equol are similar across different species, though there may be small differences.
Studies on rats and monkeys (using blood and urine samples) show that the main byproduct of S-equol is S-equol-4′-glucuronide, which makes up most of the conjugated byproducts, with smaller amounts of monosulfate compounds and mixed glucuronide-sulfate conjugates [11]. In vitro studies (done in labs) using human, rat, and monkey liver cells (hepatocytes) confirm this too—almost all S-equol is processed within 180 minutes of exposure, and the main byproduct in all three species is S-equol-4′-glucuronide, showing the processing pathway is very similar across species [11].
Another study on ovariectomized rats (rats without ovaries, used as a model for estrogen deficiency) found that when racemic equol (a mix of S and R forms) is taken by mouth, glucuronide conjugates are still the main byproducts—suggesting estrogen levels do not change how S-equol is processed [12].
However, a semi-quantitative study on two people who make S-equol found slightly different results: their blood samples showed the main byproduct was S-equol-7-glucuronide-4′-sulfate, followed by S-equol-7-glucuronide and S-equol-7-sulfate [13]. This differs from the liver cell studies, possibly because lab conditions (in vitro) are different from the human body (in vivo), or because people process S-equol differently [13].
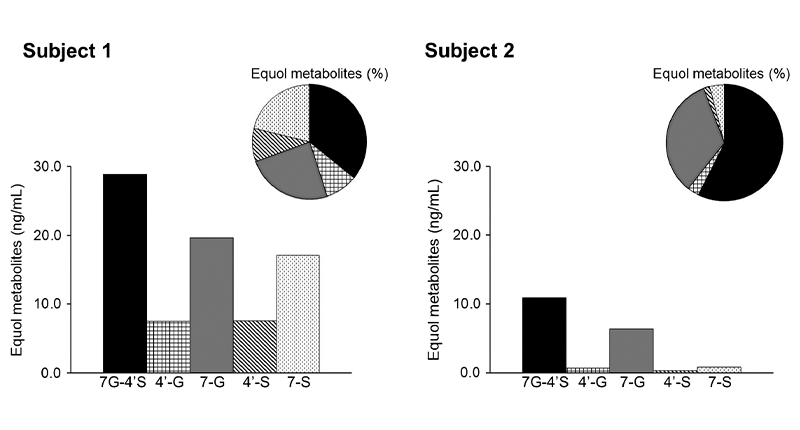
Figure 3. Concentration Proportion of Equol Metabolites in Plasma of Two Subjects [15]
S-Equol Pharmacokinetics: How the Body Processes It
Absorption
Absorption is when a substance enters the bloodstream from where it is taken (like the mouth). For oral S-equol, this mostly happens in the gastrointestinal tract—mainly the small intestine—and how well it is absorbed directly affects how much S-equol gets into the bloodstream, a key factor in its biological effectiveness. Research consistently shows that S-equol is very well absorbed when taken by mouth, much easier than its precursor daidzein—one of the reasons it is considered the most active form of soy isoflavones.
Time to maximum concentration (Tmax): A Phase I clinical study in Japan found that S-equol is absorbed quickly after oral administration. After single doses of 10–320 mg or multiple doses of 10–160 mg (twice a day), the median Tmax (time to reach the highest blood level) is 1.5 to 3 hours [14]. A review of existing data reported a slightly wider range of 1–6 hours [15], thought to be due to differences in supplement formulation (e.g., tablets vs. capsules), dosage, and whether it is taken with food—food can slightly slow absorption but does not affect overall absorption.
In a specific study of postmenopausal women, a 30 mg commercial S-equol supplement (SE5-OH, which contains S-equol, daidzein, genistein, and glycine) reached peak blood concentration in about 1 hour [16], showing that formulation greatly affects absorption speed.
Plasma concentration: A study of non-Equol producers (people who cannot make S-equol on their own) found that S-equol levels in the blood rose significantly within 45 minutes of a single 6 mg dose, from a baseline of 2–4 ng/mL to 121–137 ng/mL [17]. This confirms even small exogenous doses can reach biologically active levels. Notably, male subjects had lower circulating S-equol levels than postmenopausal women in the same study [18], which may affect effective dosing for men.
Oral bioavailability: Clinical data shows S-equol has a high apparent bioavailability of about 82%—much higher than daidzein (30–40%) [16]. In postmenopausal women who took 10 mg or 30 mg of SE5-OH, about 82% of the administered S-equol (in both free and conjugated forms) was recovered in urine over 0–48 hours [16], confirming its high absorption rate. Another study estimated the gastrointestinal absorption rate is around 80% [17], further supporting its excellent oral bioavailability.
Distribution
After absorption into the bloodstream, S-equol is carried by blood proteins to various tissues and organs. Its distribution determines if it can reach target organs (like bones, brain, or prostate) and exert its effects, as well as its elimination rate. In vitro cell uptake experiments initially predicted about 49.7% of circulating S-equol is free (unbound), but in vivo studies show it binds strongly to plasma proteins in all tested species: 96.5% in rats, 97.3% in monkeys, and 96.1% in humans [20], meaning only a small fraction (0.4%–5.0% in rats and monkeys) is free [20].
Even so, the free S-equol concentration is still 100 times higher than the Ki value (dissociation constant) needed to bind to estrogen receptor β (ERβ) [21], meaning it is sufficient to exert biological activity by binding to ERβ and regulating downstream processes.
Clinical studies report an apparent volume of distribution of 96.2 L for S-equol [22]—much larger than the average adult’s total body fluid volume (about 42 L)—suggesting it has good lipid solubility and can penetrate tissues effectively. This tissue penetration is not gender-dependent, so S-equol works equally well in men and women (though doses may need adjustment based on blood levels).
Specific tissue distribution studies are limited, but one study on white men who consumed soy long-term found equol accumulates in the prostate in an inverse gradient manner: its concentration in prostatic fluid was 4 to 13 times higher than in plasma, with S-equol accounting for 12.7 times the plasma concentration [23–25]. This is thought to relate to high ERβ expression in prostate epithelial cells and S-equol’s ability to specifically bind to 5α-dihydrotestosterone (DHT), a hormone involved in prostate health.
Additionally, computer simulations and in vitro models show S-equol has high blood-brain barrier (BBB) permeability (with a -log Pe value of approximately 39.7), making it easier to enter the central nervous system than daidzein [26]—crucial for its potential neuroprotective effects.
Metabolism
As mentioned earlier, S-equol is mainly metabolized in the liver through phase II conjugation reactions—specifically glucuronidation and sulfation. These reactions add glucuronic acid or sulfate groups to the hydroxyl groups at the 4′ and 7 positions, increasing water solubility and making it easier to excrete [11]. Importantly, these conjugated metabolites usually lose biological activity, as conjugation modifies the structure needed to bind to ERβ. The liver is the primary metabolic site, but intestinal bacteria play a minor role in deconjugation during enterohepatic circulation, which can prolong S-equol’s half-life.
Excretion
Urine is the main route of elimination for S-equol and its metabolites. Multiple studies show the elimination half-life of S-equol in healthy adults is approximately 7–8 hours (biological half-life of 8 hours) [14], supporting a twice-daily (every 12 hours) dosing schedule to maintain stable plasma concentrations and reduce peak-trough fluctuations.
According to pharmacokinetic principles, a drug typically reaches steady-state plasma concentrations after 4–5 half-lives, but S-equol reaches steady-state in about 24 hours [14]—faster than expected, likely due to enterohepatic circulation recycling some S-equol back into the bloodstream.
The apparent clearance rate of S-equol is 8.73 L/h, with a renal clearance rate ranging from 60.2 to 869 mL/h [14,22,29]. For comparison, daidzein’s apparent clearance rate is 30.09 L/h, meaning S-equol is eliminated more slowly—contributing to its longer duration of action.
Notably, S-equol undergoes enterohepatic circulation: in female rats administered daidzein or S-equol, S-equol concentration in bile was hundreds to thousands of times higher than in serum [30]. Besides the primary absorption peak 1–2 hours after administration, secondary plasma peaks often occur at 4–5 hours and 8–9 hours [12,28]—corresponding to gallbladder emptying and intestinal reabsorption of deconjugated S-equol, strong evidence of enterohepatic circulation.
S-Equol Safety and Toxicology: Is It Safe?
Overall, S-equol has an excellent safety profile. It has been extensively studied in animal toxicology and human clinical trials, with no evidence of serious adverse effects. Some S-equol raw materials have obtained GRAS (Generally Recognized As Safe) certification and are classified as GHS Category 5/Unclassified [29,30]. In genetic toxicity tests meeting ICH guidelines for new drug registration, S-equol has shown no genotoxicity [30].
Clinical Studies
To date, 33 clinical studies of S-equol have been registered, including 2 dedicated safety studies using 99.5% pure chemically synthesized S-equol from Ausio Pharmaceuticals [14]. A single ascending dose study (10–320 mg) and a multiple ascending dose study (10–160 mg, twice daily) both showed all participants tolerated S-equol well: no drug-related serious adverse events (AEs) occurred, no AEs required intervention or study drug discontinuation, and there were no significant changes in routine serum chemistry, complete blood counts, electrocardiograms, or blood pressure [14].
Daiichi Sankyo (Japan) has focused on postmenopausal non-Equol producers in multiple studies, confirming its product SE5-OH (containing S-equol, daidzein, genistein, and glycine) has good safety. It does not cause serious treatment-related AEs affecting the hormonal environment (sex hormones, gonadotropins, and thyroid hormones), indicating S-equol does not disrupt endogenous hormone levels or pose additional hormonal risks [31,32].
In a study focusing on reproductive organs, reported AEs included vaginal discharge, breast tenderness, and vaginal bleeding—but their incidence was not significantly different from the placebo group [33], confirming S-equol does not harm reproductive organs.
Animal Studies
Two safety studies included in SE5-OH’s GRAS certification showed it has good safety in acute and subchronic toxicity tests, with no significant toxic effects. SE5-OH also showed no genotoxicity or mutagenicity, and its No Observed Adverse Effect Level (NOAEL) was 2000 mg/kg/day—the highest dose tested [34].
Acute toxicity study: The oral LD50 (lethal dose for 50% of test animals) of SE5-OH is greater than 4000 mg/kg. At 4000 mg/kg, no deaths, treatment-related changes in clinical signs, food consumption, or body weight were observed [34].
91-day subchronic toxicity study: No deaths occurred in either control or treatment groups. Rat body weight gain was the same across groups, with no significant pathological findings. There were no significant differences in water consumption, organ weights, or behavior, and minor statistical changes in male rat white blood cell count and prothrombin time were within normal physiological ranges and not toxicologically significant. Urine and clinical chemistry results also showed no significant group differences [34].
Genetic toxicity studies: 1. Bacterial reverse mutation test: SE5-OH showed no mutagenicity in Salmonella typhimurium strains (TA98, TA100, TA1535, TA1537) and Escherichia coli strain WP2uvrA, with or without S9 metabolic activation [35]. 2. Chromosome aberration test: SE5-OH did not induce chromosome aberrations in Chinese hamster lung cells at concentrations up to 3000 μg/mL, with or without metabolic activation [35]. 3. Micronucleus test: SE5-OH did not induce an increase in micronucleated polychromatic erythrocytes when administered to Sprague-Dawley rats by gavage at doses up to 4000 mg/kg, twice daily for 2 consecutive days [35].
Reproductive and developmental toxicity studies: SE5-OH had a NOAEL of 1000 mg/kg/day (equivalent to 6.5 mg/kg/day S-equol) for both male and female rats. During the developmental toxicity phase, no effects on embryos or fetuses were found at any tested dose, and the NOAEL for developmental toxicity was 2000 mg/kg/day (equivalent to 13 mg/kg/day S-equol) [35].
Additional Safety Studies
In animal models of breast cancer, S-equol did not stimulate the growth of estrogen-dependent human breast cancer MCF-7 cells transplanted into ovariectomized mice [36]. In another model, it did not stimulate the growth of mammary tumors induced by the chemical carcinogen 7,12-dimethylbenz[a]anthracene (DMBA), though it also did not show chemopreventive effects [37].
A correlation analysis of human data showed S-equol production status is not associated with increased breast cancer risk in postmenopausal U.S. women [38]. Additionally, in MCF-7 (estrogen-dependent) and MDA-MB-231 (triple-negative) breast cancer cells, S-equol did not significantly upregulate transporter expression. Instead, it inhibited breast cancer resistance protein (BCRP) activity and enhanced cancer cell sensitivity to BCRP substrates, indicating it does not increase chemotherapy resistance [39].
References
[1] Marrian G F, Haslewood G A. Equol, a new inactive phenol isolated from the ketohydroxyoestrin fraction of mares' urine [J]. The Biochemical Journal, 1932, 26(4): 1227–32.
[2] Gardana C, Canzi E, Simonetti P. The role of diet in the metabolism of daidzein by human faecal microbiota sampled from Italian volunteers [J]. The Journal of Nutritional Biochemistry, 2009, 20(12): 940–7.
[3] Setchell K D, Clerici C. Equol: history, chemistry, and formation [J]. The Journal of Nutrition, 2010, 140(7): 1355S–62S.
[4] Gardana C, Canzi E, Simonetti P. R(-)-O-desmethylangolensin is the main enantiomeric form of daidzein metabolite produced by human in vitro and in vivo [J]. Journal of Chromatography B, 2014, 953–954: 30–7.
[5] Mayo B, Vázquez L, Flórez A B. Equol: A bacterial metabolite from the daidzein isoflavone and its presumed beneficial health effects [J]. Nutrients, 2019, 11(9).
[6] Janning P, Schuhmacher U S, Upmeier A, et al. Toxicokinetics of the phytoestrogen daidzein in female DA/Han rats [J]. Archives of Toxicology, 2000, 74(8): 421–30.
[7] Křížová L, Dadáková K, Kašparovská J, et al. Isoflavones [J]. Molecules, 2019, 24(6).
[8] 赵晓佳, 李易聪, 王秀伶. 大豆异黄酮微生物转化研究进展 [J]. 微生物学报, 2020, 60(02): 211–26.
[9] Zhang T, Wang B, Wang C, et al. In vivo and in vitro mechanisms of equol synthesis and key influencing factors: A critical review [J]. Nutrients, 2025, 17(21).
[10] Wang Q, Spenkelink B, Boonpawa R, et al. Use of physiologically based kinetic modeling to predict rat gut microbial metabolism of daidzein to S-equol [J]. Molecular Nutrition & Food Research, 2020, 64(6): e1900912.
[11] Schwen R J, Nguyen L, Jackson R L. Elucidation of the metabolic pathway of S-equol in rat, monkey and man [J]. Food and Chemical Toxicology, 2012, 50(6): 2074–83.
[12] Legette L L, Prasain J, King J, et al. Pharmacokinetics of equol in ovariectomized rats [J]. Journal of Agricultural and Food Chemistry, 2014, 62(6): 1294–300.
[13] Obara A, Kinoshita M, Hosoda K, et al. Identification of equol metabolites in human plasma [J]. Pharmacology Research & Perspectives, 2019, 7(3): e00478.
[14] Jackson R L, Greiwe J S, Desai P B, et al. Pharmacokinetics of S-equol [J]. 2011, 18(2): 185–93.
[15] Hernandez G, Zhao L, Franke A A, et al. Pharmacokinetics and safety of phytoSERM formulation [J]. Menopause, 2018, 25(2): 191–6.
[16] Setchell K D, Zhao X, Shoaf S E, et al. Pharmacokinetics of S-equol in postmenopausal women [J]. The Journal of Nutrition, 2009, 139(11): 2037–43.
[17] Stewart K L, Lephart E D. Overview of BPH and equol [J]. International Journal of Molecular Sciences, 2023, 24(6).
[18] Hazim S, Curtis P J, Schär M Y, et al. Effects of equol on arterial stiffness in men [J]. The American Journal of Clinical Nutrition, 2016, 103(3): 694–702.
[19] Nagel S C, Vom Saal F S, Welshons W V. Free fraction of estrogens in serum [J]. Proceedings of the Society for Experimental Biology and Medicine, 1998, 217(3): 300–9.
[20] Schwen R J, Nguyen L, Plomley J B, et al. Toxicokinetics of S-equol [J]. Food and Chemical Toxicology, 2012, 50(5): 1741–8.
[21] Setchell K D, Clerici C, Lephart E D, et al. S-equol as ERβ ligand [J]. The American Journal of Clinical Nutrition, 2005, 81(5): 1072–9.
[22] Setchell K D, Zhao X, Jha P, et al. Pharmacokinetics of equol isomers [J]. The American Journal of Clinical Nutrition, 2009, 90(4): 1029–37.
[23] Hedlund T E, Maroni P D, Ferucci P G, et al. Isoflavone metabolism in prostate [J]. The Journal of Nutrition, 2005, 135(6): 1400–6.
[24] Lund T D, Blake C, Bu L, et al. Equol and prostate health [J]. Reproductive Biology and Endocrinology, 2011, 9: 4.
[25] Sekikawa A, Weaver A, Mroz K, et al. S-equol and bone health [J]. 2025, 17(24): 3962.
[26] Johnson S L, Kirk R D, Dasilva N A, et al. BBB permeability of polyphenols [J]. Metabolites, 2019, 9(4).
[28] Fujitani M, Mizushige T, Bhattarai K, et al. Enterohepatic circulation of S-equol [J]. Biomedical Research, 2019, 40(3): 97–105.
[29] Anandakumar S R, Handral M, Seekallu S. Safety assessment of S-equol [J]. Toxicology Reports, 2024, 13: 101823.
[30] Schwen R, Jackson R, Proudlock R. Genotoxicity assessment of S-equol [J]. Food and Chemical Toxicology, 2010, 48(12): 3481–5.
[31] Ishiwata N, Melby M K, Mizuno S, et al. Equol and menopausal symptoms [J]. Menopause, 2009, 16(1): 141–8.
[32] Aso T, Uchiyama S, Matsumura Y, et al. S-equol supplementation in postmenopausal women [J]. Journal of Women’s Health, 2012, 21(1): 92–100.
[33] Oyama A, Ueno T, Uchiyama S, et al. Effects of S-equol on skin aging [J]. Menopause, 2012, 19(2): 202–10.
[34] Yee S, Burdock G A, Kurata Y, et al. Toxicity of SE5-OH [J]. Food and Chemical Toxicology, 2008, 46(8): 2713–20.
[35] Matulka R A, Matsuura I, Uesugi T, et al. Developmental effects of SE5-OH [J]. Journal of Toxicology, 2009.
[36] Ju Y H, Fultz J, Allred K F, et al. Equol and breast cancer growth [J]. Carcinogenesis, 2006, 27(4): 856–63.
[37] Brown N M, Belles C A, Lindley S L, et al. Chemopreventive action of equol [J]. Carcinogenesis, 2010, 31(5): 886–93.
[38] Virk-Baker M K, Barnes S, Krontiras H, et al. Equol and breast cancer risk [J]. Nutrition Research, 2014, 34(2): 116–25.
[39] Rigalli J P, Scholz P N, Tocchetti G N, et al. Equol and BCRP inhibition [J]. European Journal of Nutrition, 2019, 58(1): 139–50.




































 Time:Mar 05, 2026
Time:Mar 05, 2026
 Share:
Share:










 +1(425)866-9043
+1(425)866-9043
 sales@bonerge.com
sales@bonerge.com