
Many people notice that as they age, their sleep becomes lighter, they wake up earlier, and daytime energy fades more quickly.
These changes are often dismissed as a natural part of aging. However, growing evidence suggests that they reflect a deeper disruption of the body’s biological rhythm — the core timing system that coordinates physiological processes throughout the day.
At the center of this system is a master clock located in the suprachiasmatic nucleus (SCN), together with a network of peripheral clocks distributed throughout the body. Together, these clocks precisely coordinate the periodic expression of approximately 40% of genes.
This molecular timing mechanism is based on a transcription–translation feedback loop (TTFL), involving core components such as the CLOCK/BMAL1 heterodimer and clock genes including Period (PER1/2/3) and Cryptochrome (CRY1/2). These gene products undergo phosphorylation and changes in subcellular localization, forming a self-sustaining oscillatory cycle of approximately 24 hours.
Recent studies have revealed a bidirectional regulatory relationship between biological rhythm systems and cellular aging. On the one hand, clock genes influence the aging process by regulating longevity-associated pathways such as SIRT1 and AMPK. On the other hand, oxidative stress and mitochondrial dysfunction associated with aging disrupt the oscillatory properties of clock genes.
This vicious cycle leads to circadian disturbances commonly observed in elderly individuals, such as phase advances and amplitude attenuation. Research suggests that, compared to younger individuals, elderly individuals may experience sleep onset and wake-up times 1-2 hours earlier. This phase advance is not merely a behavioral change (such as going to bed earlier) but reflects intrinsic changes in the biological clock itself.
Beyond phase shifting, the "dampening" of rhythmic amplitude is another significant age-related change. This means that physiological indicators such as body temperature and hormone levels fluctuate less prominently.
For instance, compared to younger individuals, elderly individuals exhibit a ~14% reduction in lipid circadian rhythm amplitude, a 50% decrease in melatonin secretion amplitude in individuals over 80 years old, and a ~0.3°C reduction in body temperature fluctuation. The weakening of circadian rhythms is believed to increase susceptibility to various age-related diseases, including diabetes, dyslipidemia, and neurodegenerative disorders.

Figure 1: Compared to young individuals (blue line), many rhythmic amplitudes are weakened in elderly individuals (red line).In some cases, the peak of rhythms also shifts forward. [Image source: DOI: 10.1172/JCI90328]
Molecular Decoupling of Biological Rhythms in Aging: Urolithin A Boosts Rhythmic Amplitude
To explore the molecular basis of aging-related circadian disruption, the research team at Nagasaki University established a senescence model using TIG-3 cells. This system provided an ideal platform for analyzing circadian alterations associated with cellular aging.
Using real-time bioluminescence monitoring, researchers observed that the oscillatory amplitude of the Bmal1 gene in senescent cells decreased by 67%, while the circadian period lengthened by 2.3 hours. This phenotype closely mirrors the rhythm attenuation observed in aging organisms, validating the cellular model.
The study then examined the effects of ellagic acid (EA) and urolithin A (UA) on circadian clock gene expression. Results showed that UA significantly enhanced the rhythmic amplitude of Bmal1 in senescent cells (by approximately fourfold). A similar enhancement was also observed in proliferative cells, suggesting that UA’s effect on circadian regulation may not be limited to aging populations.
In contrast, EA itself exhibited no significant influence on circadian properties in either senescent or proliferative cells. Further analysis revealed that other EA-derived metabolites, including urolithin B and urolithin C, also displayed amplitude-enhancing potential, although their effects were weaker than that of UA.

Figure 2: Metabolites of EA, rather than EA itself, exhibit potential in enhancing circadian rhythmic amplitude (Figure A), with UA showing the most significant effect among the EA metabolites (Figure B).
To further elucidate the mechanism underlying UA-induced enhancement of Bmal1 rhythmic amplitude, researchers employed a luciferase fusion protein detection system. UA treatment significantly reduced the stability of the Per2-luc protein, shortening its half-life from 3 hours to 1.7 hours. This finding suggests that UA promotes Per2 protein degradation.
Given that Per2 protein stability is partly regulated by its acetylation status, and that SIRT1-mediated deacetylation facilitates Per2 degradation via the ubiquitin–proteasome system, the researchers next examined whether UA affected NAD⁺ levels and SIRT1 expression.
Although UA did not significantly alter intracellular NAD⁺ levels in senescent TIG-3 cells, SIRT1 protein levels showed an increasing trend following UA treatment, while SIRT1 mRNA levels decreased by approximately 50%. These results indicate that UA likely regulates SIRT1 through post-transcriptional mechanisms, such as enhanced protein stability or translation efficiency, rather than direct transcriptional activation.
Senescent cells accumulate with age and are considered a major contributor to aging-related diseases due to their disrupted biorhythmic state. The ability of UA to modulate the circadian properties of senescent cells may therefore contribute to restoring rhythmic amplitude across a wide range of physiological processes governed by biological clocks. This property positions UA as a promising nutritional candidate for mitigating biorhythmic dysfunction.
Dual Mechanism of Urolithin A in Reversing the Aging-Related Circadian Phenotype
Nagasaki University’s latest findings further clarify the rhythm-resynchronizing properties of urolithin A. This study is the first to demonstrate that UA restores circadian oscillations in senescent cells through a dual mechanism involving both the SIRT1–PER2 axis and chromatin remodeling.
At the molecular level, UA shortened the half-life of Per2 protein from 3 hours to 1.7 hours, increasing its degradation rate by 76%. Despite a 50% reduction in SIRT1 mRNA expression, SIRT1 protein levels increased by 1.8-fold, indicating enhanced protein stability or translational efficiency.
Beyond protein turnover, UA also exerted a profound effect on chromatin architecture. ATAC-seq analysis revealed that UA increased chromatin accessibility in the Bmal1 regulatory region by 3.2-fold. Concurrently, H3K27ac modification levels at CLOCK-binding sites increased by 2.7-fold, creating a chromatin environment more favorable for rhythmic gene transcription.
Metabolomic analysis further showed that UA restored succinate levels in senescent cells, increasing them by 2.1-fold. As a key tricarboxylic acid cycle intermediate, succinate has been shown to enhance circadian gene amplitude via activation of the succinate receptor SUCNR1. Together, these findings outline a metabolic–epigenetic–circadian regulatory network underlying UA’s multi-target effects.
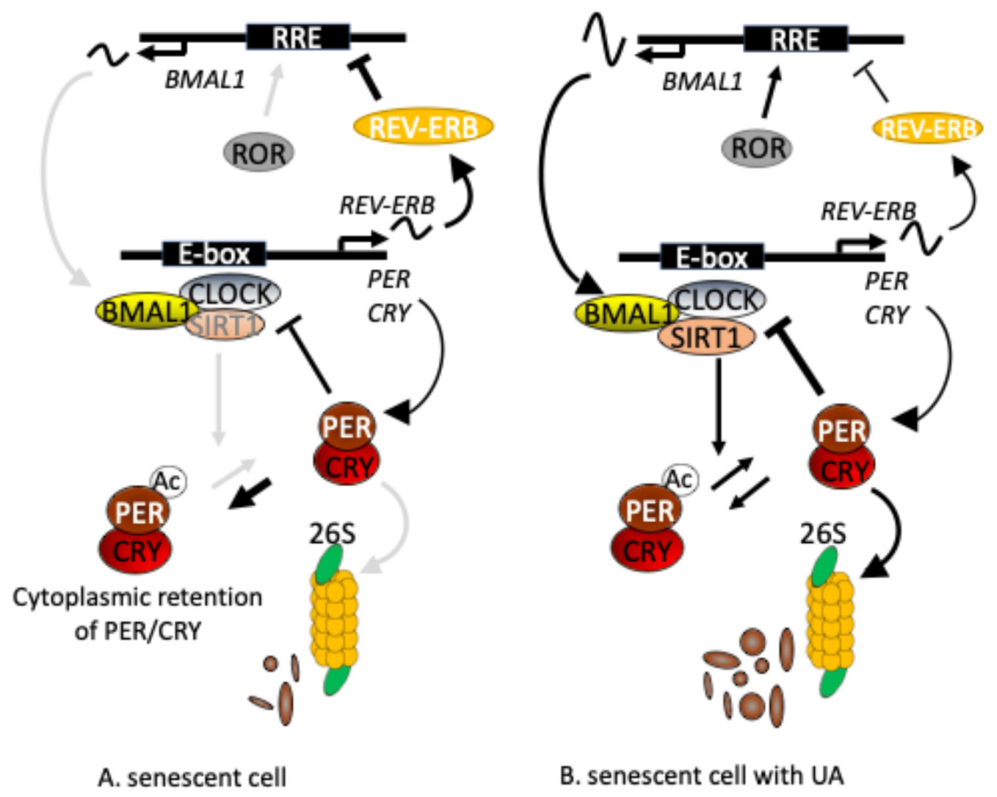
Figure 3: Mechanistically, UA may stabilize SIRT1 protein, enhance its deacetylation activity, thereby promoting PER2 deacetylation and degradation, ultimately suppressing CLOCK/BMAL1 activity and enhancing Bmal1 rhythmic expression.
Urolithin A and Circadian Regulation: A Potential "Dark Horse" for Boosting Daytime Energy?
Many aspects of human physiology are governed by the biological clock, including sleep–wake cycles, hormone secretion, metabolism, and immune function. The emerging evidence surrounding UA highlights its potential to reverse aging-related circadian phenotypes by restoring rhythmic gene amplitude.
Consistent with this concept, previous studies have shown that dietary supplementation with oak extract—metabolized into EA and urolithins—increased the activity/rest ratio in aged mice, an effect not observed in young mice. These findings suggest that UA may selectively enhance circadian robustness in aging organisms.
Taken together, UA’s ability to enhance rhythmicity may extend to a broad range of physiological processes influenced by circadian regulation, including metabolic efficiency, sleep quality, and daytime energy levels. As mechanistic understanding deepens, UA may emerge as a nutritional strategy for optimizing biological clock function and improving quality of life.
It is also noteworthy that ellagic acid, the dietary precursor of UA, is abundant in foods such as pomegranate and walnuts. However, human conversion efficiency varies widely (0–50%), highlighting the potential need for personalized supplementation strategies or microbiome-based optimization approaches.
Finally, it is important to recognize that most current evidence on UA and circadian regulation is derived from cellular and animal studies. While these findings are promising, they do not fully represent human physiology. Therefore, further investment in well-designed human clinical trials is essential to validate UA’s safety, efficacy, optimal dosage, and potential side effects, thereby establishing a robust scientific foundation for its future applications in healthcare and medical nutrition.
References
[1] Nutrients2025, 17(1), 20.
[2] Commun Biol 6, 756 (2023).
[3] J Physiol Sci 66, 367–374 (2016).
[4] Neurol Clin. 2019 May 29;37(3):487–504.
[5] Cell. 2008 Jul 25;134(2):317-28.




































 Time:Aug 18, 2025
Time:Aug 18, 2025
 Share:
Share:










 +1(425)866-9043
+1(425)866-9043
 sales@bonerge.com
sales@bonerge.com