For many women in their 30s and beyond, long working hours, chronic stress, poor sleep, and delayed childbearing have become common.
Over time, these lifestyle factors may lower ovarian reserve. This can appear as irregular cycles, lower AMH levels, and earlier signs of aging in reproduction.
Recent clinical and preclinical studies continue to explore Urolithin A benefits in female fertility and ovarian reserve biology. Researchers increasingly discuss Urolithin A, a naturally occurring postbiotic compound, among female fertility supplements designed to support long-term reproductive health.
Now, new preclinical data reveal how Urolithin A may support ovarian reserve and female fertility at the molecular level.

What is Urolithin A
Urolithin A ( UA ) is a natural postbiotic compound produced by gut bacteria from precursors found in certain foods (pomegranates, walnuts, etc. ), and it has attracted research interest for its potential anti-aging and health benefits.
Only about 40% of people can naturally convert dietary precursors into meaningful levels of urolithin A, making direct supplementation an option for others to achieve beneficial levels. 1
Clinical studies have demonstrated that supplementing with Urolithin A:
Regulating Circadian Rhythms & Improving Sleep Quality 2
Reducing Fatigue from Sleep Deprivation 3
Enhances NAD+/NADH Recycling & Boosts Cellular Energy 4
With its proven efficacy and safety, Urolithin A is now being studied for a wide range of aging-related concerns beyond sleep health, including ovarian function & female Fertility.

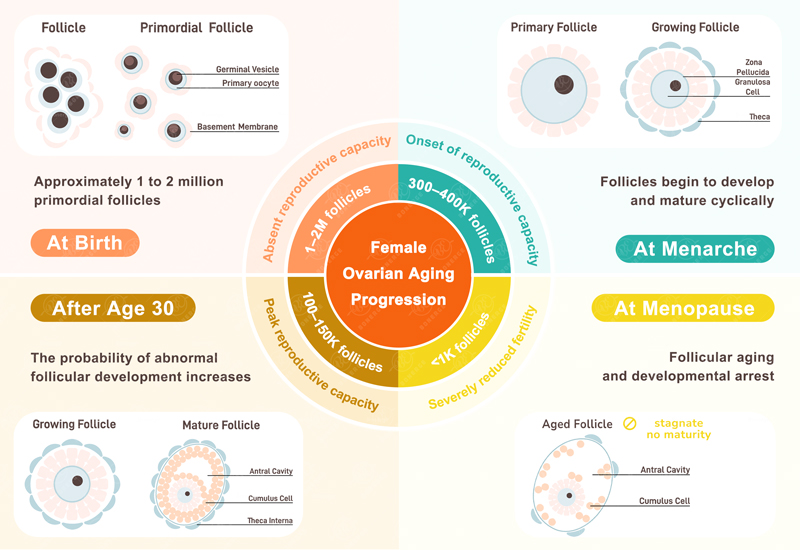
Understanding Ovarian Reserve and Primordial Follicle Dormancy
Every woman has about one million primordial follicles at birth. These follicles form a fixed and non-renewable ovarian reserve. Each primordial follicle houses a potential egg, serving as the starting point for life conception. These follicles do not develop simultaneously. 5
Throughout a woman’s reproductive life, most primordial follicles remain dormant. Only a small number activate, mature, and release during each menstrual cycle.
This special "conservation" mechanism helps a woman’s fertility last for many years. However, if this mechanism does not work right, follicles can run out too fast. This can lead to an early decline in ovarian reserve function.
Thus, maintaining the dormant state of primordial follicles is the core key to extending a woman’s fertility window.
New Animal Data on Urolithin A and Female Fertility
A 2025 animal study published in Biology provides new insights into how Urolithin A may support ovarian reserve and female fertility. 6
Researchers focused on a key biological pathway involved in early follicle activation: PI3K/Akt signaling.

Ovarian Reserve Decline and PI3K/Akt Signaling
Researchers first looked at past studies on ovarian reserve decline.
They confirmed that abnormal PI3K/Akt signaling plays a key role in early follicle activation. When this pathway stays too active, primordial follicles wake up too early. Over time, this leads to ovarian reserve depletion.
Previous research also showed that chemotherapy drugs such as cyclophosphamide cause DNA damage in the ovary. This damage not only triggers cell death but also speeds up follicle loss.
Based on this background, researchers asked two simple questions:
Can blocking PI3K/Akt signaling keep follicles dormant?
Can it reduce chemotherapy-related ovarian damage?
To answer these questions, they studied the urolithin A benefits in ovarian protection models.
Urolithin A Maintains Follicle Dormancy
In mouse ovary culture models, urolithin A supplements showed clear protective effects.
Researchers observed:
Fewer growing follicles
Lower levels of oocyte growth markers
Reduced support cell growth
No increase in cell death
These findings suggest that urolithin A supplement benefits include helping follicles stay in a resting state.
Keeping follicles dormant is important for long-term ovarian reserve health. This is one of the key urolithin A benefits related to female reproductive aging.
Urolithin A Protects Against Chemotherapy-Induced Damage
Chemotherapy can quickly damage the ovarian reserve.
In both lab and animal models, urolithin A supplements helped protect the ovary.
Researchers found:
Higher numbers of preserved primordial follicles
Lower levels of cell death signals
Reduced activation of DNA damage pathways
These results suggest that urolithin A supplement benefits may include protection against chemotherapy-related fertility loss.
This makes urolithin A a promising candidate among supplements for fertility for females who face ovarian stress.
How Urolithin A Works
Researchers then studied how it works inside the ovary.
They found that urolithin A reduced activity in the PI3K/Akt pathway. This pathway controls follicle activation.
When its activity decreased:
Follicles were less likely to activate too early
Cell stress signals were lower
DNA damage responses were reduced
This dual effect helps explain the fertility-supporting urolithin A benefits seen in the models.
Clinical Research Trends in Ovarian Reserve and Female Fertility
In recent years, clinical research has focused more on ovarian reserve. A key indicator of long-term reproductive potential exists. Clinicians commonly evaluate biomarkers such as AMH levels, antral follicle count, and cycle regularity to better understand reproductive aging.
At the same time, researchers are exploring how dietary supplements and naturally occurring compounds may influence cellular processes that affect ovarian biology. Rather than targeting hormone levels directly, many emerging strategies investigate how signaling pathways, mitochondrial health, and oxidative balance contribute to overall reproductive system function.
As clinical trials change, more people are interested in methods that keep ovarian reserve stable over time. This research direction shows a larger trend toward precision reproductive health. Here, people view long-term resilience as important as short-term results.
Beyond a Single Compound: A Multi-Pathway active ingredient Strategy
While Urolithin A plays a central regulatory role, multiple related pathways influence ovarian biology. A broader, multi-pathway strategy may include:
Clean: Supporting a Healthier Ovarian Environment
Fisetin has senolytic properties and may help reduce senescent cells. These cells can release inflammation-related signals that impair follicle development. 7
Regulate: Maintaining Metabolic and Autophagic Balance
Urolithin A not only helps protect the ovarian follicle reserve 6, but also promotes cellular autophagy, supporting the biological rhythm of oocytes. This contributes to maintaining normal metabolic function in oocytes and provides essential support for the development of chromosomally normal eggs with strong developmental potential.
Protect: Reinforcing Antioxidant Defense
L-Ergothioneine builds up in tissues with many mitochondria. It may help reduce stress from oxidation. This is important because oocytes are sensitive to damage from oxidation. 8
Rather than replacing hormonal pathways, such active compounds aim to support ovarian health at the cellular level.
In the broader category of supplements for female fertility, manufacturers increasingly position active compounds alongside prenatal vitamins, fatty acids, and other dietary supplements. Emerging research highlights the importance of cellular resilience and immune health in supporting long-term reproductive health.
Formulation Considerations for Female Fertility Supplements
As interest in female fertility supplements continues to grow, formulation strategy becomes increasingly important. Ingredient developers must consider not only efficacy-related pathways but also stability, bioavailability, and long-term safety profiles.
In ovarian reserve support, compounds that affect cell signaling, mitochondrial function, and oxidative balance can help. These can work alongside traditional nutrients like prenatal vitamins and fatty acids. A well-designed formulation often integrates multiple active ingredients that address different aspects of reproductive health while maintaining appropriate daily doses.
Manufacturers and brand owners are moving toward mechanism-based reproductive support. This change shows a wider market demand for precision nutrition and science-backed products. This trend shows that clinically studied active compounds are becoming more important in women's reproductive health supplements.

Why Long-Term Fertility Support Matters in Reproductive Planning
For women over 30 who want to have children in the future, long-term support is important for keeping their ovaries healthy.
Instead of focusing only on short-term outcomes, research increasingly emphasizes the need to support ovarian reserve, cellular resilience, and metabolic balance. These biological systems help sustain reproductive health over time.
Such approaches do not replace medical treatment. Rather, they complement broader reproductive care by supporting ovarian function in the long term.
Science-Driven Approaches to Ovarian Reserve Support
As reproductive science advances, interest continues to grow around non-hormonal, mechanism-based strategies that may support ovarian reserve and cellular integrity.
Current evidence shows that Urolithin A may help female fertility in the long run. It may also support the basic function of ovarian reserve.
Clinical trials are still developing. However, interest is growing in how Urolithin A supplements may boost female fertility. These supplements may work through specific mechanisms instead of using high doses or sudden hormonal treatments.
For ingredient developers and formulation strategists, these findings show that active compounds are becoming more important for ovarian health. This is part of the larger area of women’s reproductive support.
Bonerge remains committed to exploring science-backed pathways that contribute to long-term reproductive resilience.
Reference:
[1] Singh A, et al. 2022. Cell Reports Medicine. 3(5):100633. doi:10.1016/j.xcrm.2022.100633. PMID: 35600264.
[2] Du Y, et al. 2024. International Journal of Molecular Sciences. 25(13):7021. doi:10.3390/ijms25137021. PMID: 39064706.
[3] Zhu H, et al. 2024. Food & Function. 15(6):2703–2716. doi:10.1039/D3FO04567A. PMID: 38468112.
[4] Ghosh N, et al. 2020. Scientific Reports. 10:20184. doi:10.1038/s41598-020-76564-7. PMID: 33214689.
[5] Zhu Z, et al. 2022. Aging Cell. 21(3):e13535. doi:10.1111/acel.13535. PMID: 35132962.
[6] Wang W, et al. 2025. Biology. 14(7):612. doi:10.3390/biology14070612. PMID: 40723388.
[7] Hense JD, et al. 2024. Nature Reviews Endocrinology. 20(4):231–246. doi:10.1038/s41574-024-00873-9. PMID: 38422119.
[8] D’Amico D, et al. 2021. Trends in Molecular Medicine. 27(7):687–699. doi:10.1016/j.molmed.2021.04.003. PMID: 33933428.
[9] Borodina I, et al. 2020. Antioxidants. 9(11):1030. doi:10.3390/antiox9111030. PMID: 33153160.




































 Time:Feb 13, 2026
Time:Feb 13, 2026
 Share:
Share:











 +1(425)866-9043
+1(425)866-9043
 sales@bonerge.com
sales@bonerge.com