The liver handles a remarkable workload. It processes nutrients, filters toxins, produces proteins, and manages both lipid and glucose metabolism. Modern diets, sedentary habits, and rising rates of obesity and type 2 diabetes are putting it under growing strain. The result is the world's most common chronic liver disease: metabolic dysfunction-associated steatotic liver disease, or MASLD.
Doctors formerly called this condition nonalcoholic fatty liver disease (NAFLD). In 2023, an international expert panel introduced the new name to reflect what actually drives it. The cause is not simply the absence of alcohol. It is a cluster of metabolic issues: insulin resistance, lipid imbalance, and excess body fat [1].
This renaming shifts focus toward root causes. That focus is exactly where researchers and supplement formulators are now looking.
Studies show that around 38% of adults globally have MASLD today [2]. Projections suggest this figure could exceed 55% by 2040 [2]. In high-income Western countries, modelling data indicate MASLD prevalence in men could approach 64% by 2030 [3].
MASLD is already the leading indication for liver transplant among women in the United States [2]. Left unaddressed, it contributes to liver damage, liver injury, and in advanced cases, liver failure.
The search for effective liver health supplements is active. Researchers want compounds that act on liver metabolism at a specific, known level. In May 2025, a team at Naval Medical University in China published findings that stand out in this field.
After screening 2,543 natural compounds, the team identified dihydroberberine (DHB) as the single most potent activator of a critical liver regulatory molecule. The compound showed no cellular toxicity [4].
This article covers that discovery. It compares DHB to other liver support supplements and explains why formulators and ingredient researchers are paying attention.
Understanding MASLD: The Iron and Fat Connection
From Fatty Liver to MASLD
Doctors diagnose MASLD when liver cells accumulate excess fat alongside at least one metabolic risk factor. These include elevated body mass index (BMI), impaired fasting glucose, type 2 diabetes, high triglycerides, or high blood pressure [1].
Low or zero alcohol intake does not protect against fatty liver disease. If metabolic health is impaired, liver fat can still accumulate. MASLD is a metabolic condition, not an alcohol-related one.
MASLD progresses along a spectrum. Simple fatty liver can advance to metabolic dysfunction-associated steatohepatitis (MASH) -- active inflammation of the liver. From there, the risk of fibrosis, cirrhosis, and liver cancer rises. Studies estimate the annual rate of progression from MASLD to MASH at 7-35% [5].
Iron Overload: A Compounding Risk Factor
Fat accumulation defines MASLD. But research points to a second factor that compounds liver damage: excess iron. Iron is essential for oxygen transport and energy production. When hepatic iron rises beyond what the liver can regulate, it becomes a significant source of oxidative stress. This is more common in people with obesity, insulin resistance, or metabolic syndrome.
Excess iron generates reactive oxygen species (ROS). These damage liver cells and drive lipid peroxidation.
Iron also interacts with polyunsaturated fatty acids in a fat-loaded liver. Together, these factors create conditions for ferroptosis -- a form of iron-dependent, oxidative cell death. This iron-lipid cycle accelerates liver injury beyond what either factor causes on its own [4].
miR-122: The Liver's Master Regulatory Switch
At the centre of this cycle sits a microRNA called miR-122. It accounts for around 52% of all microRNA in human liver cells [4]. miR-122 controls genes for fatty acid synthesis, lipid oxidation, cholesterol metabolism, and immune system activity in the liver.
When iron overload advances, miR-122 levels fall. The liver then loses control over lipid production.
Lipogenic genes become overexpressed. Polyunsaturated fatty acid levels rise. Oxidative damage escalates. In the presence of excess iron, lipid peroxidation and ferroptosis follow. This worsens MASLD at the cellular level [4].
Restoring miR-122 is a logical target for liver support. The question is: which compound can do this effectively and safely?
The 2025 Study: Screening 2,543 Compounds
The study appeared in Pharmacological Research in May 2025. The team at Naval Medical University in Shanghai, China, had one goal: find natural compounds that raise miR-122 expression [4].
The team built a luciferase reporter assay. This tool works like a signal light: when miR-122 activity rises, the signal brightens. The researchers used this system to screen 2,543 natural compounds. For each one, they tested whether it raised miR-122 levels without harming cell viability.
Shortlisted compounds went through secondary dose-response testing and cytotoxicity checks. Compounds with significant miR-122 upregulation at safe concentrations then moved to in vivo testing. The researchers used a mouse model of MASLD. The mice were fed a high-iron diet to induce the condition.

Figure 1. 2025 Pharmacological Research study identifying DHB as a top miR-122 upregulator for MASLD [4]
Dihydroberberine: The Top-Ranked Compound
From 2,543 candidates, dihydroberberine ranked first. It showed the strongest miR-122 upregulation in the luciferase reporter system. It also maintained full cell viability at active concentrations. This combination set it apart from many otherwise promising compounds [4].
In the high-iron-diet MASLD mouse model, DHB produced the following outcomes [4]:
Restoration of miR-122 expression: DHB reversed the iron overload-driven decline in hepatic miR-122. Levels returned toward those seen in healthy controls.
Reduction of liver fat: DHB reduced hepatic triglyceride content and total lipid accumulation, confirmed by Oil Red O staining.
Reduction of oxidative damage: Lipid peroxidation markers including malondialdehyde (MDA) and lipid peroxides (LPO) fell with DHB supplementation.
Improvement of liver injury markers: ALT and AST are standard clinical indicators of liver cell damage. Both dropped significantly in the DHB group.
Reduction of liver fibrosis: Histological analysis showed reduced fibrotic deposits in DHB-treated animals.
Restoration of lipid-related gene expression: DHB suppressed overexpressed lipogenic genes (Aacs, Fads2, and cytochrome P450 family members), restoring normal miR-122-driven regulation.
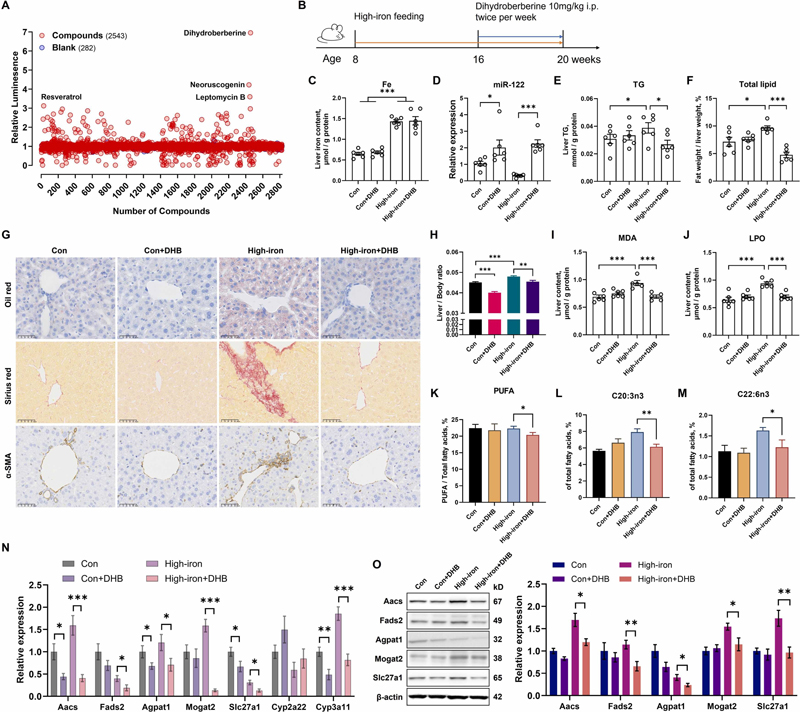
Panel A: Among 2543 compounds, dihydroberberine exhibited the most potent regulatory effect on miR‑122.
Panel C: Hepatic iron was markedly elevated in the high‑iron group and reduced by DHB treatment.
Panel D: Iron overload sharply decreased miR‑122 expression, which was restored to near‑normal by DHB.
Panels E–H: Iron overload induced hepatic triglyceride and total lipid accumulation, which were alleviated by DHB.
Panels I–J: DHB reduced iron‑overload‑driven increases in oxidative stress markers MDA and LPO.
Panels K–M: DHB corrected iron‑induced fatty acid imbalance by lowering polyunsaturated fatty acids and restoring beneficial fatty acid profiles.
Panels N–O: DHB normalized the dysregulated expression of lipogenic genes and proteins caused by iron overload. [4]
What the Mechanism Tells Us
The study maps a clear pathway. Iron overload suppresses miR-122. This releases control over lipogenic gene networks. Fat accumulates. In the presence of excess iron, lipid peroxidation and ferroptosis follow.
DHB breaks this cycle at its regulatory root. By restoring miR-122, DHB re-engages the liver's own control over lipid metabolism. It reduces the substrate available for oxidation and reduces downstream liver cell injury [4].
This mechanism is distinct from broad antioxidant or anti-inflammatory pathways. DHB targets a single regulatory switch that governs a wide network of hepatic processes. That is why its effects in this model extended across multiple biomarkers rather than just one.
Common Liver Health Supplements: Where the Field Stands
Understanding DHB's position starts with a look at the most widely used liver health supplements today. These dietary supplements set the current standard in the category.
Milk Thistle (Silymarin)
Milk thistle ranks among the most studied liver health supplements in history. Researchers extract silymarin -- its active compound -- from the seeds of Silybum marianum.
Silymarin works through several mechanisms. It scavenges free radicals, inhibits lipid peroxidation, reduces inflammation, and produces antifibrotic effects through suppression of hepatic stellate cell activation [6].
A 2023 systematic review covered 29 RCTs and 3,846 participants. It found that silymarin reduced liver enzyme levels in 65.5% of studies. Results were most consistent in nonalcoholic fatty liver disease [7].
Mayo Clinic guidelines rate its hepatoprotective use as Grade B -- good scientific evidence [8]. Standard dosing is 140 mg three times daily. The main limitation is bioavailability. Silymarin's active compounds are poorly water-soluble, and standard formulations show modest, variable absorption.
Some practitioners combine milk thistle with a vitamin E supplement for liver support. Vitamin E has shown benefits in certain NAFLD patient groups.
TUDCA (Tauroursodeoxycholic Acid)
TUDCA is a water-soluble bile acid with well-established cytoprotective properties. In the liver, it acts as a chemical chaperone. It reduces endoplasmic reticulum (ER) stress. ER stress is a key pathway in liver cell injury under metabolic stress conditions.
TUDCA has shown efficacy in reducing elevated liver enzymes and may support insulin signalling. Researchers frequently cite it in the context of drug-induced liver toxicity.
TUDCA also appears in MASLD studies. Large-scale double-blind placebo-controlled trial data remain more limited than for silymarin. Research dosing typically ranges from 500 to 1,750 mg per day.
N-Acetylcysteine (NAC)
NAC is a precursor to glutathione -- the liver's primary endogenous antioxidant. It replenishes intracellular glutathione stores, supporting the liver's ability to neutralise reactive oxygen species. NAC is best established for acetaminophen-induced liver injury, where it is the standard of care.
In MASLD and metabolic liver support, NAC shows some evidence for reducing oxidative stress markers and improving liver enzymes. Its effects on fat accumulation or fibrosis are less clearly defined. Side effects are generally mild and dose-dependent.
Berberine: Metabolic Benefits, Bioavailability Limits

Berberine (BBR) is a plant alkaloid with a long history in traditional Chinese medicine. It has attracted significant research interest for its metabolic effects: improvements in blood glucose, insulin sensitivity, lipid profiles, and gut microbiota composition.
For liver health, berberine has shown promise in preclinical studies. Some clinical trials report reductions in liver enzymes and hepatic fat in NAFLD/MASLD populations. Results vary across studies.
A 2026 clinical trial in JAMA Network Open puts those findings in context [9]. The multicenter, double-blind placebo-controlled RCT enrolled 337 diabetes-free adults with obesity and MASLD across 11 hospitals.
Participants received berberine at 1 g/day or placebo for six months. Neither visceral fat nor liver fat content changed significantly in the berberine group -- despite high medication adherence [9].
Berberine did produce modest but significant reductions in LDL-C, apoB, and high-sensitivity CRP. These findings point to cardiometabolic benefit. The primary liver outcome, however, was null [9].
The explanation likely lies in pharmacokinetics. Oral berberine bioavailability falls below 1% due to poor intestinal absorption and high first-pass metabolism [10]. At achievable oral doses, liver tissue may not receive meaningful concentrations consistently. This limits liver-specific results even when systemic lipid effects are present.
Dihydroberberine vs. Berberine: A Closer Comparison
DHB and BBR are structurally related. DHB is a reduced form of berberine. Intestinal bacteria produce it naturally when they partially reduce ingested berberine.
Once absorbed, DHB converts back to berberine in systemic circulation, where it exerts its biological effects. This gut-liver cycle means DHB delivers berberine to the body more efficiently than oral berberine itself.
Bioavailability
The key advantage of DHB is absorption. Moon and colleagues ran a 2021 randomised crossover pilot study [10]. Subjects took either 500 mg berberine, 100 mg dihydroberberine, or 200 mg dihydroberberine. The study then measured plasma berberine levels in each case.
Both doses of DHB produced significantly higher berberine AUC values over a two-hour window. A 100 mg dose of DHB delivered more plasma berberine than five times the dose of standard berberine. The study concluded that DHB produces significantly greater plasma berberine concentrations at substantially lower doses [10].
This was a small pilot study -- five healthy males. Treat the findings as preliminary pharmacokinetic evidence, not definitive dose comparison data. Larger studies will be needed to fully assess DHB's absorption profile across different populations and conditions.
Dosing and Side Effects
Standard clinical berberine dosing runs 500 mg two to three times daily -- 1,000 to 1,500 mg per day in total. At these high doses, people commonly report gastrointestinal side effects: diarrhoea, constipation, and bloating. These reflect the high local concentration of unabsorbed berberine in the gut.
DHB's improved absorption means lower doses may achieve equivalent or greater systemic levels. Less unabsorbed compound in the intestine may translate to fewer side effects and better tolerability [10].
Mechanistic Distinction
The 2025 Naval Medical University research adds a specific mechanistic argument for DHB in liver health. In the high-throughput screen, DHB ranked first for miR-122 upregulation -- not berberine [4].
Does this reflect a direct effect of DHB? Or of the berberine it converts to? Or of the combination? Researchers have not yet established the answer. Further study is needed.
The data show that DHB is the active form circulating in plasma after either compound is taken. This form has measurable activity on a critical liver regulatory pathway.
Taken together, DHB emerges as a more efficient delivery vehicle for berberine's hepatic effects. On top of that, it brings a specific mechanism tied to the iron overload-MASLD axis. Standard berberine has not yet demonstrated this in clinical studies.
Dihydroberberine as a Liver Health Supplement Ingredient
For brands, formulators, and ingredient researchers, DHB presents a strong case as a next-generation liver support supplement ingredient. The combination of mechanistic evidence, bioavailability data, and a clean safety profile is compelling.
Here are the key properties that make DHB relevant for product development:
Defined molecular mechanism: DHB's liver-relevant activity centres on miR-122 upregulation. This is a specific, measurable pathway. It sits upstream of lipid metabolism and oxidative stress cascades. Evidence-based formulation increasingly values this kind of precision. It sets DHB apart from broad-spectrum antioxidants.
Lower effective dose: Preliminary pharmacokinetic data suggest DHB achieves greater systemic berberine exposure at substantially lower doses than standard berberine. This has practical implications for capsule count, formulation flexibility, and consumer compliance.
Fewer side effects: Better absorption means less unabsorbed compound reaches the lower gut. This potentially reduces the gastrointestinal side effects that limit berberine adherence at high doses.
Safety profile: DHB showed no cytotoxicity at effective concentrations in the Naval Medical University screening -- a foundational requirement for any dietary supplement ingredient.
Additive formulation potential: DHB's mechanism complements established liver health supplements. Pairing DHB with milk thistle (silymarin), for example, addresses both the miR-122/iron axis and classical antioxidant-hepatoprotective pathways. This combination supports liver health through two distinct routes.
Growing research base: The 2025 high-throughput study and ongoing DHB metabolic research are expanding the evidence base. This supports both regulatory substantiation and consumer communication around liver support claims.
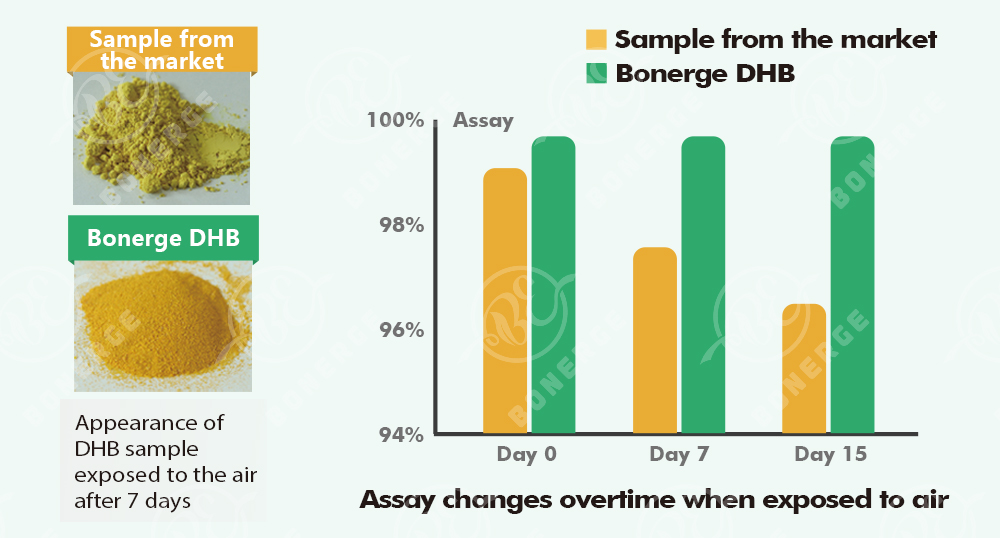
Bonerge's proprietary Dihydroberberine has a unique crystalline structure. Bonerge developed this form to solve the stability and density challenges that have historically limited DHB in commercial applications. The crystalline form achieves a bulk density above 0.6 g/ml. It also offers improved resistance to moisture absorption and degradation -- both significant considerations for manufacturers working with this ingredient at scale.
Conclusion
Research has clarified how iron and miR-122 work together to damage the liver. It has also produced a clear candidate from thousands of screened compounds. Together, these findings open a new direction for MASLD support.
Through a rigorous screen of 2,543 natural compounds, dihydroberberine emerged as the single most effective upregulator of miR-122. Iron overload suppresses miR-122. When that happens, a damaging metabolic cascade follows in the liver.
DHB restores miR-122 safely and at a defined mechanistic level. That sets it apart from most liver health supplements on the market.
Bonerge's Dihydroberberine is the only DHB ingredient on the market with a unique crystalline structure. This crystalline form achieves a bulk density above 0.6 g/ml -- a specification not available in conventional DHB. Through a proprietary manufacturing process, Bonerge addresses the three main stability challenges of standard dihydroberberine: poor physical stability, moisture absorption, and degradation over time.
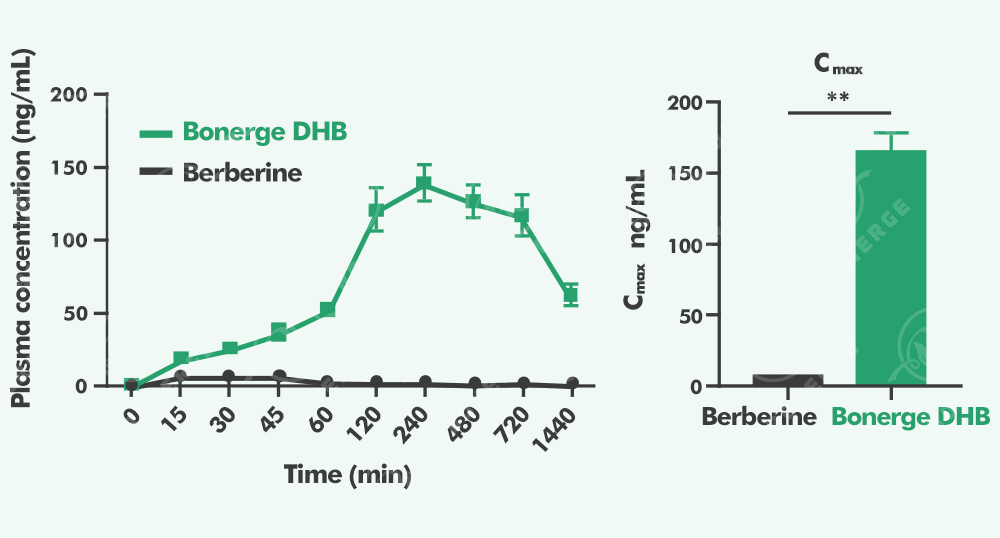
The crystalline structure also significantly enhances bioavailability. Pharmacokinetic data show that Bonerge's Dihydroberberine achieves 54 times the bioavailability of standard berberine. Bonerge continues to optimise quality parameters for this ingredient, improve its bioavailability profile, and explore its effects through ongoing animal and clinical research.
Research in this area is still growing. As pharmacokinetic data mature and longer-duration human trials emerge, the evidence base for DHB in liver health supplement formulations will grow stronger.
For more information on Bonerge's Dihydroberberine ingredient and its applications in liver health and metabolic formulations, contact our ingredient science team.

References:
[1] Rinella ME, Lazarus JV, Ratziu V, et al. A multisociety Delphi consensus statement on new fatty liver disease nomenclature. Hepatology. 2023;78(6):1966-1986. doi:10.1097/HEP.0000000000000520 -- VERIFIED.
[2] Younossi ZM, Kalligeros M, Henry L. Epidemiology of metabolic dysfunction-associated steatotic liver disease. Clin Mol Hepatol. 2025;31(Suppl):S32-S50. doi:10.3350/cmh.2024.0431 -- VERIFIED: 38% adult prevalence and >55% by 2040 confirmed from abstract.
[3] Global burden of adult non-alcoholic fatty liver disease: systematic review and meta-analysis. Translational Gastroenterology and Hepatology. 2024. -- NOTE: ~64% Western male MASLD prevalence by 2030 is a modelling projection. Presented as such in the article.
[4] Tang Y, Gao Z, Yang J, et al. Breaking the synergism of iron overload and miR-122 to rescue lipid accumulation and peroxidation in MASLD. Pharmacol Res. 2025;215:107728. doi:10.1016/j.phrs.2025.107728 -- NOTE: DOI and core findings cross-verified via multiple sources. Specific gene names (Aacs, Fads2, CYPs) and figure data should be verified against the published full text before final publication.
[5] MASLD progression rate 7-35% per year. Widely cited in MASLD literature. Primary source: Younossi ZM et al. Global epidemiology of nonalcoholic fatty liver disease. J Hepatol. 2016.
[6] Abenavoli L, Capasso R, Milic N, Capasso F. Milk thistle in liver diseases: past, present, future. Phytother Res. 2010;24(10):1423-1432. doi:10.1002/ptr.3207 -- VERIFIED.
[7] Amin MS, et al. Impact of silymarin supplements on liver enzyme levels: a systematic review. Cureus. 2023. doi:10.7759/cureus.47603 -- VERIFIED: 29 RCTs, 3,846 participants, 65.5% reduced liver enzymes.
[8] Mayo Clinic hepatoprotection Grade B classification for silymarin. Referenced via PMC9149185. Clinical guideline reference, not a primary trial.
[9] Lei L, Wang B, Zhao L, et al. Berberine and adiposity in diabetes-free individuals with obesity and MASLD: a randomized clinical trial. JAMA Netw Open. 2026;9(1):e2554152. doi:10.1001/jamanetworkopen.2025.54152 -- VERIFIED: 337 participants, 11 hospitals, 6-month double-blind placebo-controlled RCT. No significant change in VAT or liver fat at 1 g/day berberine.
[10] Moon JM, Ratliff KM, Hagele AM, Stecker RA, Mumford PW, Kerksick CM. Absorption kinetics of berberine and dihydroberberine and their impact on glycemia: a randomized, controlled, crossover pilot trial. Nutrients. 2022;14(1):124. doi:10.3390/nu14010124




































 Time:Mar 16, 2026
Time:Mar 16, 2026
 Share:
Share:











 +1(425)866-9043
+1(425)866-9043
 sales@bonerge.com
sales@bonerge.com